Cervical Plexus
The cervical plexus is a neural network formed by the ventral rami of the first four cervical spinal nerves (C1 – C4). It is situated in the upper cervical region of the neck, lying deep to the sternocleidomastoid muscle, anterior to the levator scapulae and middle scalene muscles, and posterior to the internal jugular vein and carotid sheath structures. The plexus is embedded within the prevertebral fascia, where its branches course both superficially and deeply to supply the structures of the neck and upper thoracic region.
Functionally, the cervical plexus provides motor innervation to several muscles involved in head and neck movement, sensory innervation to the skin of the neck and upper thorax, and contributes critically to respiratory function through the phrenic nerve. Through its integration of multiple cervical spinal segments, the plexus coordinates muscular stabilization of the cervical spine and transmits sensory information from the superficial tissues of the neck and shoulder region.
”Cervical Plexus”-Mikael Häggström via Wikimedia Commons. Public Domain
Plexus Branches
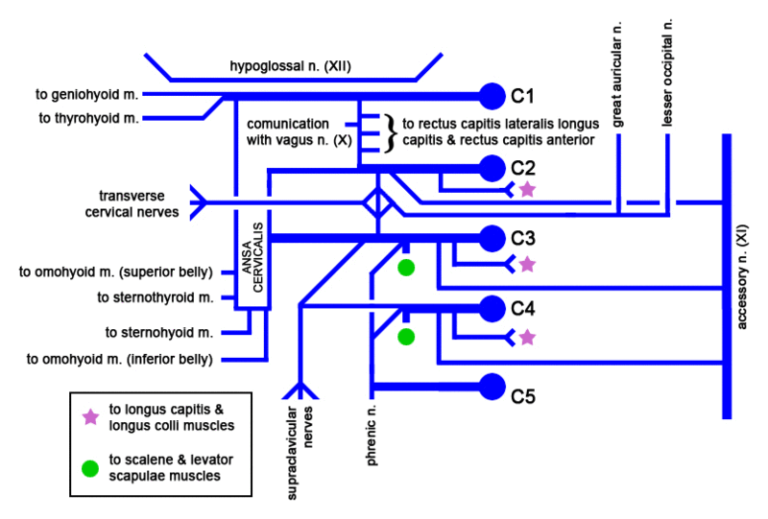
The cervical plexus (C1–C4) forms a network of motor (deep) and sensory (superficial) branches supplying the neck, upper thorax, and diaphragm. Its fibers are organized within the prevertebral fascia and distributed according to functional demands.
Motor branches innervate muscles responsible for cervical stabilization, swallowing, and respiration. The ansa cervicalis (C1–C3) supplies infrahyoid muscles, while direct branches innervate prevertebral and scalene muscles. The phrenic nerve (C3–C5) provides motor supply to the diaphragm and sensory input to thoracic structures, making it essential for respiratory function.
Sensory branches emerge at the posterior border of the sternocleidomastoid (Erb’s point) and supply the skin of the neck, lower scalp, auricle, shoulder, and upper chest via the lesser occipital, great auricular, transverse cervical, and supraclavicular nerves.
Functionally, the plexus integrates motor control and sensory feedback, supporting head posture, airway coordination, respiration, and cutaneous sensation, while providing multisegmental redundancy for stable neuromuscular function.
AI-generated illustration ( MyoAnatomy)
Motor Branches
Motor fibers from the cervical plexus supply muscles responsible for cervical spine stabilization, hyoid bone positioning, and respiratory mechanics. These motor components primarily arise from the deep branches of the plexus.
Ansa Cervicalis (C1–C3)
The ansa cervicalis is a looped nerve structure formed by contributions from C1–C3 spinal nerves. The superior root of the ansa cervicalis originates from fibers of C1 that travel briefly with the hypoglossal nerve (cranial nerve XII) before descending to join fibers from C2 and C3, forming the inferior root.
This neural loop innervates the infrahyoid muscles, including:
sternohyoid; sternothyroid; omohyoid
These muscles play a key role in stabilizing and depressing the hyoid bone and larynx during swallowing and phonation, thereby contributing to coordinated movements of the upper airway and digestive tract.
Direct Muscular Branches
Additional motor branches from the cervical plexus supply the prevertebral musculature, including
longus capitis; longus colli
These muscles are essential for cervical spine flexion and stabilization. Small branches may also contribute to the scalene muscles, which assist in neck stabilization and accessory respiratory movements by elevating the first and second ribs during forced inspiration.
Phrenic Nerve
The phrenic nerve represents the most functionally significant branch of the cervical plexus. It arises predominantly from C4, with contributions from C3 and C5.
The nerve descends vertically along the anterior surface of the anterior scalene muscle, passing deep to the prevertebral fascia and entering the thoracic cavity between the subclavian artery and vein. It continues inferiorly through the mediastinum to reach the diaphragm.
The phrenic nerve provides:
motor innervation to the diaphragm
sensory innervation to the central diaphragm, mediastinal pleura, and pericardium
Because the diaphragm is the primary muscle of inspiration, the phrenic nerve is essential for normal respiratory function. Injury to this nerve can result in hemidiaphragmatic paralysis, significantly impairing ventilation.
Sensory Branches
The cervical plexus also gives rise to several cutaneous sensory nerves that provide innervation to the skin of the neck, scalp, shoulder, and upper thoracic region. These nerves emerge at a common anatomical point along the posterior border of the sternocleidomastoid muscle, a location often referred to clinically as Erb’s point.
Direct Sensory Branches
Lesser Occipital Nerve (C2)
This nerve ascends along the posterior border of the sternocleidomastoid muscle to supply the skin of the lateral scalp posterior to the auricle and portions of the upper auricular region.
Great Auricular Nerve (C2–C3)
The great auricular nerve ascends across the sternocleidomastoid muscle toward the parotid gland and external ear. It provides sensory innervation to:
skin over the parotid region; the angle of the mandible
portions of the external ear
Transverse Cervical Nerve (C2–C3)
This nerve passes horizontally across the sternocleidomastoid muscle to supply the skin of the anterior cervical region, providing sensation to the front of the neck.
Supraclavicular Nerves (C3–C4)
The supraclavicular nerves descend into the lower neck and shoulder region, supplying the skin over the clavicle, upper chest, and superior shoulder region.
These nerves form an important sensory network that allows the central nervous system to receive cutaneous sensory information from the neck and upper thoracic region, including touch, pressure, temperature, and pain
Respiratory Control
The cervical plexus (C1–C4), with critical contribution from C3–C5, plays an essential role in respiratory mechanics through the phrenic nerve, which provides the sole motor innervation to the diaphragm.
Activation of the diaphragm generates negative intrathoracic pressure, enabling inspiration and maintaining ventilatory function. Thus, the cervical plexus is directly linked to life-sustaining respiratory control, integrating neural output with thoracic biomechanics.
Cervical Stability
Motor branches of the cervical plexus innervate prevertebral muscles, scalene muscles, and infrahyoid musculature (via ansa cervicalis, C1–C3), contributing to head stabilization, cervical flexion, and vertebral alignment.
These muscles coordinate with axial structures to maintain postural stability and controlled movement of the cervical spine, while also supporting functional activities such as swallowing and speech through hyoid stabilization.
Sensory Innervation
The cervical plexus provides cutaneous sensory innervation via its superficial branches—lesser occipital, great auricular, transverse cervical, and supraclavicular nerves—supplying the skin of the neck, lower scalp, shoulder, and upper thoracic region.
These afferent pathways transmit touch, pain, temperature, and mechanical stimuli, enabling continuous CNS monitoring of the cervical region and contributing to protective and adaptive responses.
Proprioception
Sensory fibers from the cervical plexus also convey proprioceptive input from cervical muscles and joints, which is essential for head position sense, balance, and coordinated movement.
This integration of motor output and sensory feedback forms a closed-loop control system, allowing precise regulation of cervical motion and alignment within the broader neuromuscular network.
Phrenic Nerve Injury
The most critical clinical implication of cervical plexus dysfunction involves injury to the phrenic nerve (C3–C5), which provides sole motor innervation to the diaphragm. Damage results in diaphragmatic paralysis, impairing generation of negative intrathoracic pressure.
Unilateral injury reduces ventilatory efficiency, whereas bilateral involvement leads to severe respiratory insufficiency requiring ventilatory support, underscoring the direct link between cervical plexus integrity and respiratory viability.
Cutaneous Dysfunction
Involvement of the superficial branches—lesser occipital, great auricular, transverse cervical, and supraclavicular nerves – produces sensory deficits in the neck and shoulder region. Compression, trauma, or surgical irritation may lead to pain, paresthesia, or hypersensitivity, reflecting disruption of afferent pathways.
These symptoms are commonly associated with cervical spine pathology, muscular tension, or postoperative changes, affecting regional sensory mapping and protective feedback.
Motor Disfunction
Injury to motor components, particularly the ansa cervicalis (C1–C3), compromises innervation of infrahyoid muscles, leading to impaired hyoid stabilization during swallowing and phonation.
This results in dysfunction of cervical stability and coordinated neck movement, highlighting the role of the cervical plexus in integrating motor control with functional activities such as deglutition and speech.
Cervical Relevance
Due to its proximity to major structures—carotid arteries, internal jugular vein, and scalene musculature—the cervical plexus is highly relevant in clinical procedures.
Cervical plexus block is widely used in surgeries involving the neck (e.g., carotid endarterectomy, thyroid procedures), enabling targeted sensory anesthesia. However, its anatomical location also makes it vulnerable to iatrogenic injury during surgery or trauma, necessitating precise anatomical knowledge for safe intervention.
Motor Control
Peripheral nerves provide the motor interface between the central nervous system and skeletal muscle, enabling controlled force generation through alpha motor neuron discharge and neuromuscular junction (acetylcholine-mediated) transmission. This initiates excitation–contraction coupling within muscle fibers, leading to mechanical force production.
Regulation of motor unit recruitment and rate coding determines force magnitude, timing, and gradation, allowing precise execution of movement, joint stabilization, and maintenance of posture.
Sensory Feedback
Peripheral nerves transmit continuous afferent input from musculoskeletal receptors to the CNS, forming the basis of proprioceptive control. Muscle spindles encode changes in length and velocity, Golgi tendon organs monitor tension, and joint mechanoreceptors detect position and load.
This input is processed at both spinal and supraspinal levels (including cerebellar integration), enabling real-time modulation of motor output and maintaining accuracy, coordination, and adaptability of movement.
Reflex Regulation
Peripheral nerves mediate spinal reflex circuits that provide rapid, automatic responses independent of conscious control. Reflexes such as the stretch reflex regulate baseline muscle tone and postural stability, while withdrawal reflexes protect against harmful stimuli.
These mechanisms operate through tightly regulated sensorimotor loops, ensuring immediate neuromechanical adjustment and protection against excessive mechanical stress or injury.
Functional Impact
Peripheral nerves function as a closed-loop neuromechanical system, integrating motor output, sensory feedback, and reflex activity into coordinated movement. This system maintains neuromechanical coupling, allowing efficient force transmission, balance, and adaptation to changing mechanical demands. Disruption of peripheral nerve function results in denervation, impaired excitation–contraction coupling, altered feedback gain, and loss of coordinated motor control, leading to weakness, atrophy, instability, and inefficient movement patterns.
Peripheral Nerve Injury Mechanism
Peripheral nerve dysfunction arises from disruption of axonal conduction and neural integrity, impairing bidirectional signal transmission between the CNS and musculoskeletal tissues. Common mechanisms include mechanical compression (entrapment), traumatic injury (stretch, laceration), inflammatory processes, and metabolic disorders.
These insults may cause demyelination (slowed conduction) or axonal degeneration (loss of signal transmission), leading to impaired excitation–contraction coupling and defective sensory signaling. Additionally, compromised microvascular supply via the vasa nervorum contributes to ischemic nerve injury, further exacerbating dysfunction.
Motor Dysfunction
Damage to motor fibers disrupts alpha motor neuron output, resulting in reduced or absent muscle activation. This leads to weakness, paralysis, and impaired motor unit recruitment, ultimately compromising force generation and joint stability.
Prolonged denervation results in muscle atrophy and loss of neuromuscular efficiency, transforming movement into a mechanically unstable and energetically inefficient process. Clinically, this is evident in conditions such as radial nerve injury (wrist drop), where loss of extensor function severely alters upper limb biomechanics
Sensory Loss
Sensory fiber involvement disrupts transmission of afferent signals from mechanoreceptors and nociceptors, leading to loss of proprioception, impaired coordination, and altered movement accuracy. Without adequate feedback, the CNS cannot effectively modulate motor output, resulting in ataxia and instability. Simultaneously, nerve irritation or damage may generate neuropathic pain, characterized by abnormal signaling within sensory pathways. Conditions such as carpal tunnel syndrome (median nerve compression) and sciatica (sciatic nerve irritation) illustrate how sensory dysfunction produces both pain and impaired motor control.
Clinical Impact
Peripheral nerve dysfunction represents a failure of the neuromechanical control system, disrupting the integration of motor output, sensory feedback, and reflex regulation. This leads to combined deficits in strength, coordination, and adaptive control, significantly impairing functional movement.
Systemic conditions such as peripheral neuropathy further exacerbate this by causing diffuse nerve damage and progressive functional decline. Clinically, accurate diagnosis requires understanding nerve anatomy and physiology to localize lesions, differentiate motor vs sensory deficits, and guide targeted rehabilitation or intervention strategies.
