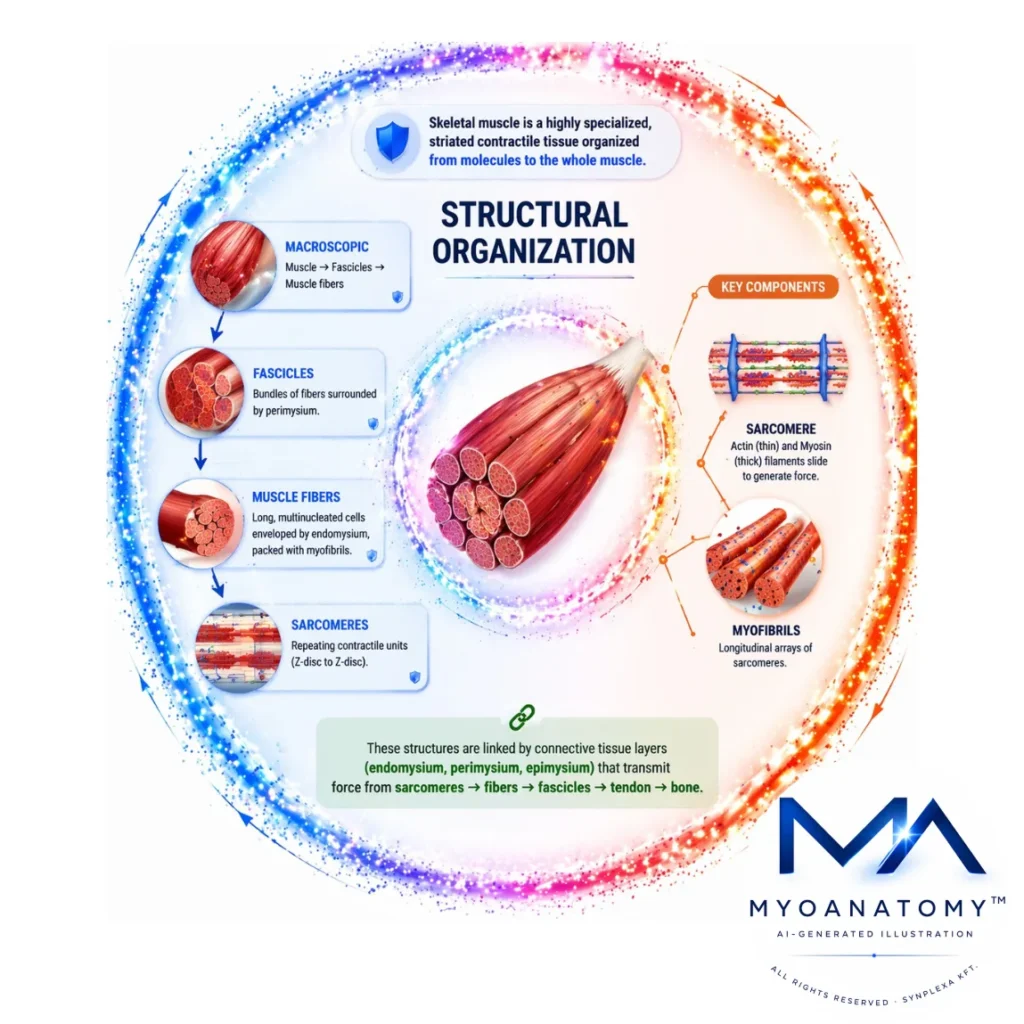
Structural Organization
Skeletal muscle is a highly specialized, striated contractile tissue organized in a precise hierarchical manner that extends from molecular components to the level of the whole muscle.
At the macroscopic level, each muscle is enclosed by the epimysium and composed of multiple fascicles, which are bundles of muscle fibers surrounded by perimysium; these fascicles define functional compartments and contribute to the directional properties of force production. Each muscle fiber is a long, multinucleated cell enveloped by endomysium and densely packed with myofibrils, which are longitudinal arrays of repeating contractile units known as sarcomeres. The sarcomere represents the fundamental functional unit of contraction, bounded by Z-discs and composed of interdigitating thin (actin) and thick (myosin) filaments whose sliding interaction underlies force generation at the molecular level.
These structural elements are not isolated but are mechanically integrated through a continuous connective tissue network – endomysium, perimysium, and epimysium – which converges into tendons, allowing the transmission of force from individual sarcomeres across fibers and fascicles to skeletal attachments. This hierarchical and connective organization ensures both longitudinal and lateral force transmission, mechanical stability during contraction, and efficient coordination of muscle activity.
SARCOMERE ORHGANIZATION
“1003 Thick and Thin Filaments” – OpenStax College, Anatomy & Physiology. Wikimedia Commons. Licensed under CC BY 4.0

“Muscle Fibers Structure” – OpenStax, Anatomy&Physiology, CNX via Wikimedia Commons.
Licensed under CC BY 4.0.
AI-Generated Illustiaton-MyoAnatomy
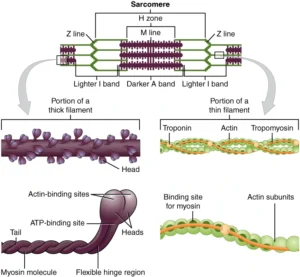
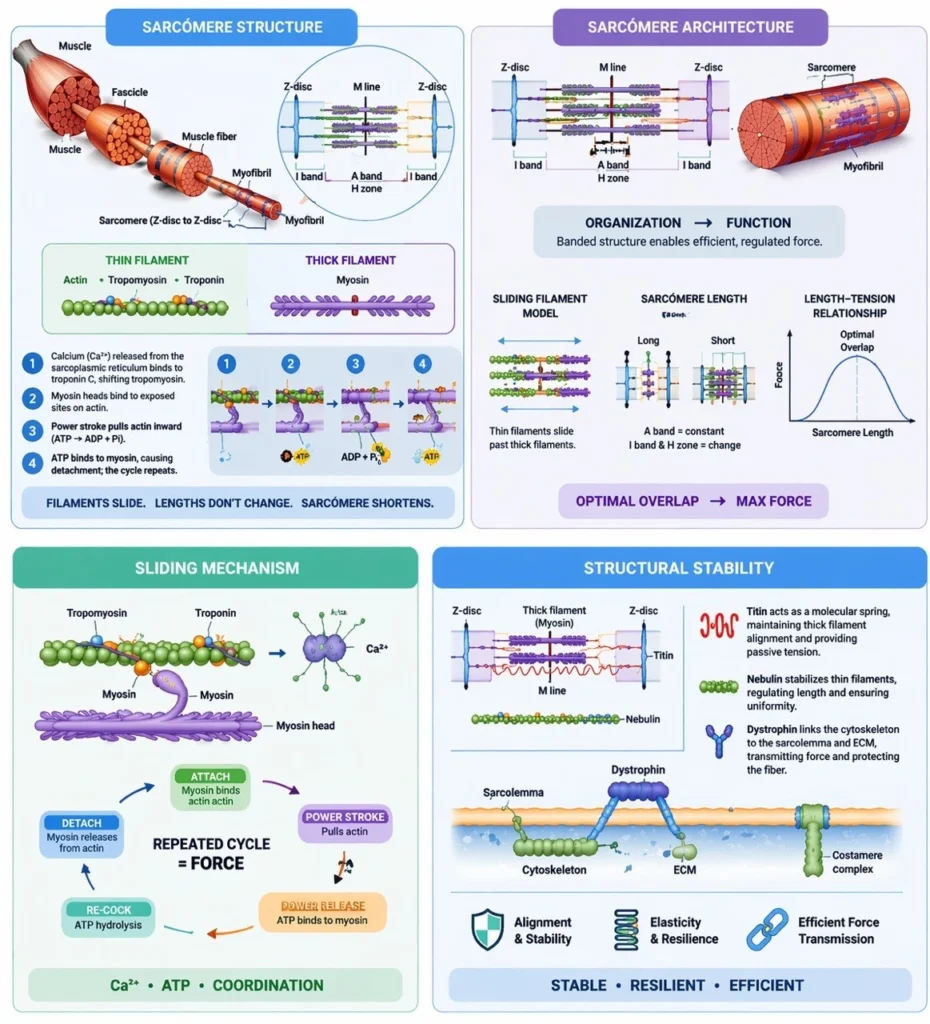
Sarcomere Structure
Skeletal muscle contraction is fundamentally dependent on the precisely regulated interaction between actin (thin filaments) and myosin (thick filaments) within myofibrils, forming an integrated contractile system supported by structural and regulatory proteins. Thin filaments consist primarily of actin, along with the regulatory proteins troponin and tropomyosin, which control access to myosin-binding sites in a calcium-dependent manner. Thick filaments are composed of myosin molecules whose heads function as ATP-dependent molecular motors, generating force through cyclic cross-bridge formation.
In addition to contractile proteins, the sarcomere contains critical structural elements, including titin, which provides passive elasticity and maintains central alignment of thick filaments, and nebulin, which stabilizes thin filament length. Other cytoskeletal proteins, such as dystrophin, anchor the contractile apparatus to the sarcolemma, facilitating force transmission to the extracellular matrix.
These components are organized into repeating units known as sarcomeres, extending from one Z-disc to the next. The Z-discs serve as anchoring sites for thin filaments and play a crucial role in maintaining structural continuity between adjacent sarcomeres. This highly ordered arrangement ensures that contractile force generated at the molecular level is efficiently transmitted along the length of the myofibril, enabling coordinated and uniform muscle contraction.
Exam Question
“Provide a detailed mechanistic account of skeletal muscle contraction at the sarcomere level, integrating the roles of actin, myosin, regulatory proteins (troponin–tropomyosin complex), and structural proteins (titin, dystrophin). Explain how these components coordinate to produce controlled, efficient force generation and ensure structural integrity during repeated contraction cycles
Sarcomere Architecture
Within each sarcomere, the precise spatial organization of filaments creates distinct structural regions that are essential for optimal force generation and contraction efficiency. The A band corresponds to the full length of the thick filaments and includes areas of overlap with thin filaments, representing the primary site of cross-bridge formation. In contrast, the I band contains only thin filaments and is bisected by the Z-disc, serving as a region of dynamic length change during contraction.
The H zone, located at the center of the A band, contains only thick filaments and diminishes as thin filaments slide inward during contraction. At the midpoint of the sarcomere lies the M line, composed of structural proteins that stabilize and align thick filaments, ensuring symmetrical force distribution.
This highly ordered geometric arrangement allows for optimal filament overlap, which is critical for maximal cross-bridge interaction and efficient force production. Changes in sarcomere length directly influence force generation, forming the basis of the length–tension relationship in muscle physiology. Thus, sarcomere architecture is not only structurally precise but functionally optimized to regulate contraction dynamics and mechanical output.
Exam Question
Analyze how the spatial organization of the sarcomere (A band, I band, H zone, M line, and Z-discs) determines the efficiency of force production. In your answer, incorporate the sliding filament mechanism and explain how variations in sarcomere length influence cross-bridge formation and the length–tension relationship
Sliding Mechanism
Skeletal muscle contraction is mediated by the sliding filament mechanism, a highly coordinated molecular process in which thin (actin) filaments slide past thick (myosin) filaments without a change in their individual lengths, resulting in sarcomere shortening. This process is initiated by Ca²⁺ release from the sarcoplasmic reticulum, which binds to troponin C, inducing a conformational change that shifts tropomyosin away from myosin-binding sites on actin.
Exposure of these binding sites allows myosin heads, energized by ATP hydrolysis, to form cross-bridges with actin. The subsequent power stroke, driven by the release of inorganic phosphate and ADP, generates force and pulls the thin filament toward the center of the sarcomere. Detachment of the myosin head requires binding of a new ATP molecule, followed by re-cocking of the head through ATP hydrolysis, enabling cyclic repetition of cross-bridge formation.
This tightly regulated cycle ensures synchronous contraction, with coordinated shortening of sarcomeres across the myofibril. Importantly, contraction results from filament sliding rather than filament shortening, preserving structural integrity while allowing efficient force generation.
Exam Question
Describe in detail the molecular sequence of events underlying the sliding filament mechanism, integrating the roles of Ca²⁺, troponin–tropomyosin regulation, ATP-dependent cross-bridge cycling, and explain how this process results in sarcomere shortening without alteration of filament length.
Structural Stability
The structural integrity of the sarcomere during repeated cycles of contraction is maintained by a network of cytoskeletal and anchoring proteins that ensure alignment, elasticity, and efficient force transmission. Titin, an elastic protein extending from the Z-disc to the thick filament, functions as a molecular spring, maintaining central positioning of myosin filaments and contributing to passive tension and recoil following stretch.
Nebulin runs along the length of thin filaments, acting as a stabilizing scaffold that regulates filament length and ensures uniformity across sarcomeres. Dystrophin links the intracellular cytoskeleton to the sarcolemma and extracellular matrix, providing structural reinforcement and facilitating the transmission of contractile force to the surrounding connective tissue while protecting the muscle fiber from mechanical stress.
Together, these proteins form an integrated stabilizing system that preserves sarcomeric alignment, prevents excessive deformation during contraction, and ensures that force generated at the molecular level is efficiently transmitted to produce coordinated macroscopic movement.
Exam Question
Explain how structural proteins such as titin, nebulin, and dystrophin contribute to the stability and functional integrity of the sarcomere during repeated contraction, and analyze how disruption of these components would affect force transmission and muscle performance
MYOFIBRIL ORHGANIZATION
“Blausen 0801 Skeletal Muscle” – BruceBlaus, Blausen Medical Communications via Wikimedia Commons. Licensed under CC BY 3.0
AI-Generated Illustiaton-MyoAnatomy
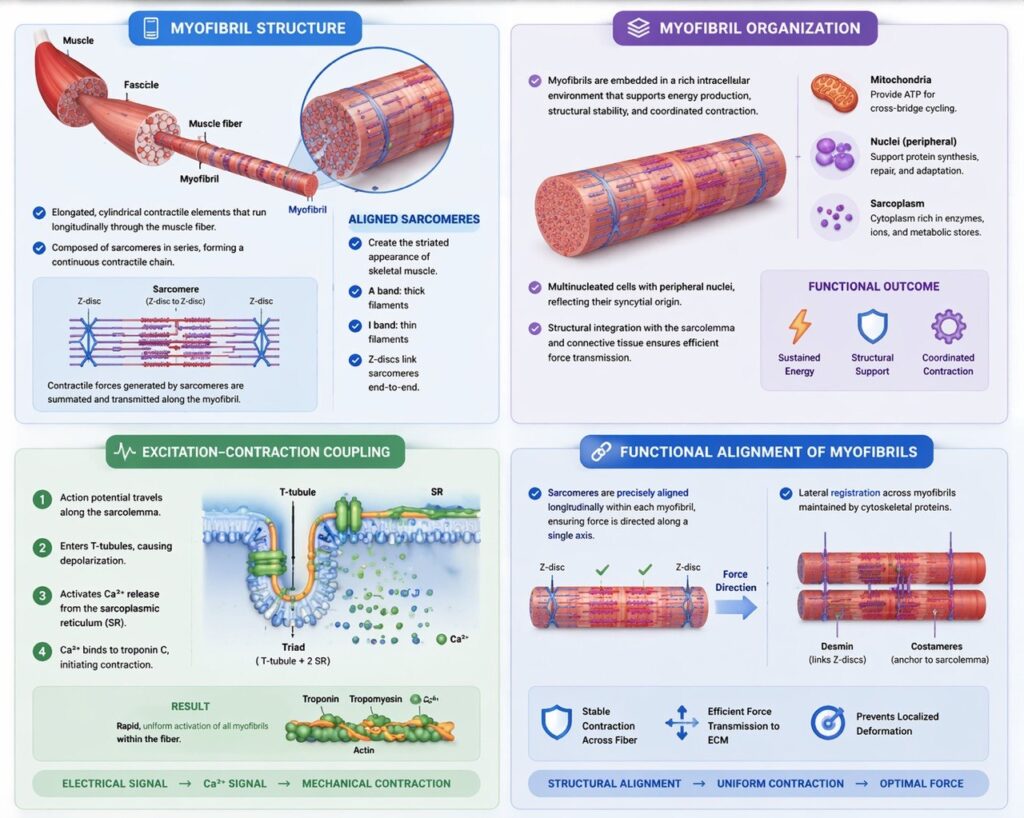
Myofibril Structure
Myofibrils are elongated, cylindrical contractile elements that extend longitudinally throughout the muscle fiber and constitute the principal functional units responsible for force generation. They are composed of sarcomeres arranged in series, forming a continuous contractile chain that spans the entire length of the cell. This serial organization ensures that contractile forces generated at the level of individual sarcomeres are summated and transmitted efficiently along the myofibril.
The precise alignment of sarcomeres produces the characteristic striated appearance of skeletal muscle, defined by the regular alternation of A bands (regions containing thick filaments) and I bands (regions containing thin filaments). This repeating pattern reflects a highly ordered internal architecture in which contractile units are mechanically linked end-to-end via Z-discs, ensuring structural continuity and coordinated shortening.
Importantly, sarcomeres do not function as isolated units; rather, they operate as components of an integrated contractile system in which force generated locally is propagated longitudinally, enabling synchronized contraction across the entire myofibril. This organization maximizes mechanical efficiency and allows precise control of muscle shortening and force output.
Exam Question
“Explain how the serial arrangement of sarcomeres within myofibrils contributes to efficient force transmission and coordinated contraction, and analyze how disruption of sarcomere alignment would affect muscle function.”
Myofibril Organization
The functional performance of myofibrils is supported by a highly integrated intracellular environment that ensures sustained energy supply, structural stability, and coordinated regulation of contraction. Mitochondria, strategically positioned between myofibrils, provide the ATP required for continuous cross-bridge cycling, thereby supporting sustained contractile activity and metabolic demand.
Skeletal muscle fibers are multinucleated, with peripherally located nuclei, reflecting their syncytial origin and enabling high levels of protein synthesis necessary for maintenance, repair, and adaptive remodeling of the contractile apparatus. This nuclear distribution optimizes transcriptional support across the extensive cytoplasmic volume of the fiber.
In addition to metabolic and nuclear organization, myofibrils are structurally integrated with cytoskeletal and membrane systems, including costameres and the sarcolemma, which facilitate force transmission from the intracellular contractile apparatus to the extracellular matrix. This coordination ensures that microscopic contractile events within myofibrils are effectively translated into macroscopic muscle contraction.
Thus, myofibril organization represents a multilevel integration of contractile, metabolic, and structural systems, enabling efficient, regulated, and sustained muscle function.
Exam Question
“Discuss how intracellular organization within skeletal muscle fibers, including mitochondrial distribution, nuclear arrangement, and cytoskeletal integration, supports sustained contraction and efficient force transmission.”
Excitation-Contraction Coupling
Rrepresents the critical physiological process by which an electrical signal is translated into mechanical contraction within skeletal muscle fibers. This process is mediated by a specialized membrane system involving the sarcolemma, transverse (T) tubules, and sarcoplasmic reticulum (SR), which together ensure rapid and coordinated activation of the contractile apparatus.
An action potential propagates along the sarcolemma and is transmitted into the interior of the muscle fiber via T-tubules, allowing depolarization to reach deep myofibrillar regions. The T-tubules are closely associated with the terminal cisternae of the SR, forming triads, which function as key sites for signal transduction. Depolarization of the T-tubule membrane activates voltage-sensitive receptors (dihydropyridine receptors), which mechanically or functionally couple to ryanodine receptors on the SR, triggering the release of Ca²⁺ into the cytosol.
The increase in intracellular Ca²⁺ concentration enables binding to troponin C, initiating conformational changes that displace tropomyosin and expose myosin-binding sites on actin, thereby activating cross-bridge cycling. This system ensures rapid, uniform activation of all myofibrils within the muscle fiber, allowing synchronized contraction.
Exam Question
Describe the sequence of events involved in excitation–contraction coupling in skeletal muscle, emphasizing the roles of the sarcolemma, T-tubules, sarcoplasmic reticulum, and Ca²⁺ signaling, and explain how this system ensures rapid and synchronized activation of contraction.”
Functional Alignment of Myofibrils
The strict longitudinal alignment of sarcomeres within each myofibril ensures that force generation is vectorially directed, allowing contraction to occur along a single axis optimized for efficient force transmission. In addition, lateral registration of sarcomeres across adjacent myofibrils is maintained by cytoskeletal proteins such as desmin and membrane-associated complexes (costameres), which mechanically couple myofibrils to each other and anchor them to the sarcolemma.
This integrated cytoskeletal network enables synchronous contraction across the entire fiber, prevents localized deformation, and facilitates effective transmission of force not only along the myofibrils but also to the sarcolemma and extracellular matrix. As a result, skeletal muscle achieves stable, uniform, and directionally controlled mechanical output at the cellular level.
Exam Question
Analyze how the longitudinal and lateral alignment of sarcomeres and myofibrils contributes to efficient force generation and transmission, and explain the roles of cytoskeletal proteins such as desmin and costameres in maintaining structural and functional integrity.”
MUSCLE FIBER ORHGANIZATION
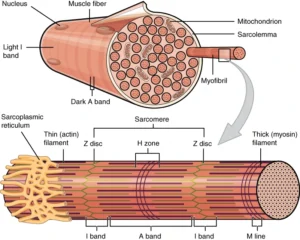
“1002 Organization of Muscle Fiber” – OpenStax Collge, Anatomy & Physiology via Wikimedia Commons. Licensed under CC BY 4.0
AI-Generated Illustiaton- MyoAnatomy
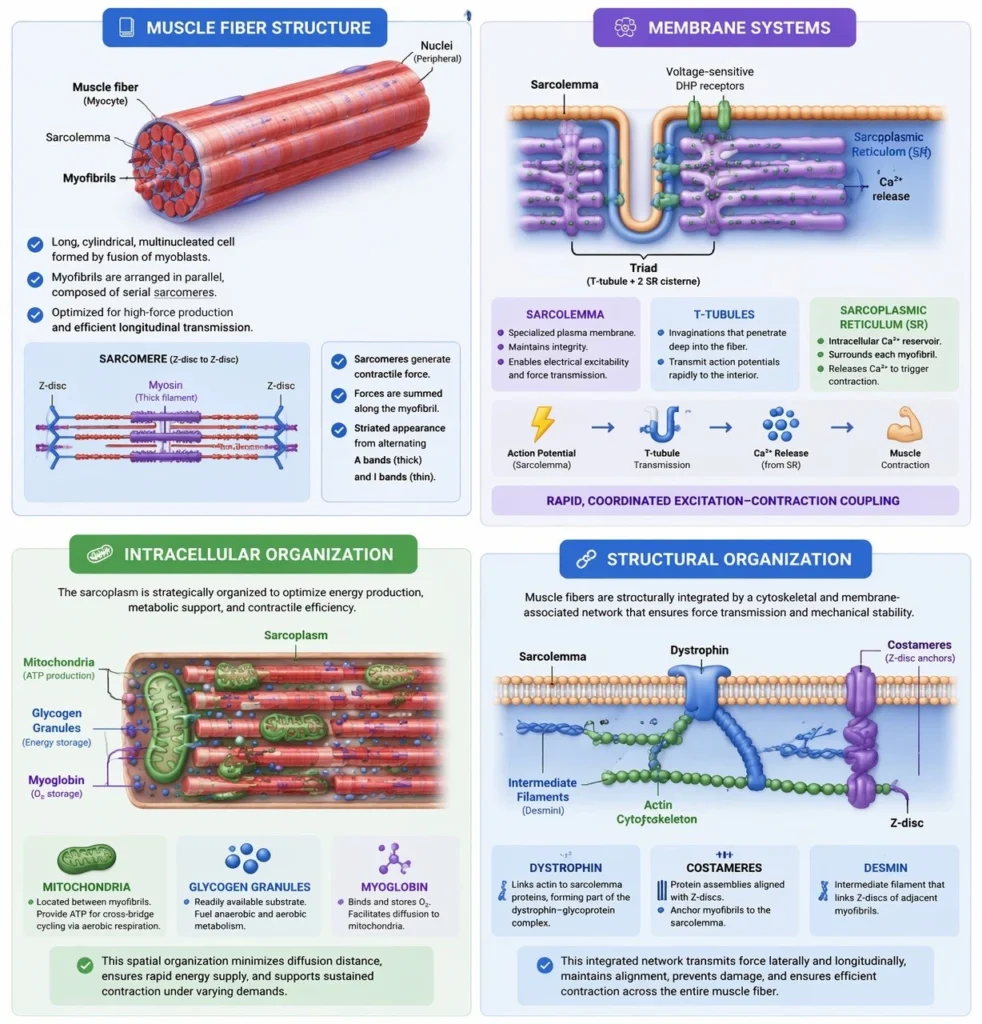
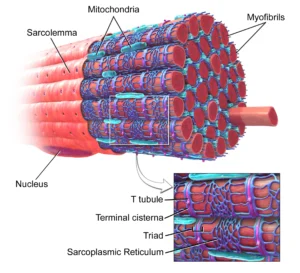
Muscle Fiber Structure
The skeletal muscle fiber (myocyte) is a long, cylindrical, multinucleated cell formed through the fusion of myoblasts during embryonic development, resulting in a syncytial structure optimized for high-force production. The cytoplasm, known as the sarcoplasm, is densely packed with myofibrils arranged in parallel along the longitudinal axis, ensuring efficient transmission of contractile force throughout the fiber.
Each muscle fiber is enclosed by the sarcolemma, a specialized plasma membrane that not only maintains cellular integrity but also plays a crucial role in electrical excitability and force transmission. The sarcolemma interfaces with the extracellular matrix via membrane-associated complexes, enabling the transfer of contractile force beyond the cell.
The presence of multiple peripherally located nuclei reflects the high metabolic and synthetic demands of skeletal muscle, allowing localized regulation of protein synthesis across the extensive cellular volume. This multinucleation supports continuous maintenance, repair, and adaptive remodeling of the contractile apparatus.
Exam Question
“Describe the structural organization of a skeletal muscle fiber, including its syncytial nature, sarcolemmal function, and nuclear distribution, and explain how these features contribute to efficient force generation and transmission.”
Membrane Systems
Skeletal muscle fibers possess highly specialized membrane systems that are essential for rapid and coordinated excitation–contraction coupling, ensuring precise temporal control of contraction. The sarcoplasmic reticulum (SR) forms an extensive intracellular network surrounding each myofibril and serves as the primary reservoir for Ca²⁺, which is required for activation of the contractile machinery.
Invaginations of the sarcolemma, known as transverse (T) tubules, penetrate deep into the muscle fiber, allowing action potentials to be rapidly transmitted from the cell surface to the interior. The close anatomical relationship between T-tubules and the SR gives rise to triads (one T-tubule flanked by two terminal cisternae of the SR), which function as key sites for coupling electrical signals to Ca²⁺ release.
Depolarization of the T-tubule membrane activates voltage-sensitive receptors that trigger Ca²⁺ release from the SR, ensuring that all myofibrils within the fiber are activated simultaneously. This system enables rapid, uniform contraction and prevents asynchronous activation that would compromise mechanical efficiency.
Thus, the membrane system represents a highly specialized conduction and signaling network that integrates electrical excitation with intracellular Ca²⁺ dynamics, ensuring synchronized and efficient muscle contraction.
Exam Question
Explain the structural organization and functional role of the sarcoplasmic reticulum and T-tubule system in skeletal muscle, and analyze how their interaction ensures rapid and synchronized excitation–contraction coupling
Intracellular Organization
The sarcoplasm of skeletal muscle fibers is highly specialized to support sustained, high-efficiency contraction, with organelles strategically organized to optimize energy delivery and metabolic support. Mitochondria are densely distributed between myofibrils, positioning ATP production in close proximity to sites of cross-bridge cycling, thereby minimizing diffusion distances and ensuring continuous energy supply during contraction.
In addition to mitochondrial density, the sarcoplasm contains glycogen granules, which serve as a readily accessible substrate for anaerobic and aerobic metabolism, and myoglobin, an oxygen-binding protein that facilitates intracellular oxygen storage and diffusion to mitochondria. Together, these components support both rapid and sustained energy demands under varying physiological conditions.
The spatial arrangement of these organelles reflects a highly optimized intracellular architecture, where metabolic systems are tightly coupled to the contractile apparatus. This organization enhances metabolic efficiency, reduces energy loss, and ensures rapid responsiveness, enabling skeletal muscle to maintain force production during prolonged or repetitive activity.
Exam Question
Discuss how the intracellular organization of skeletal muscle fibers, including mitochondrial distribution, glycogen storage, and myoglobin content, supports sustained contraction and metabolic efficiency.”
Structural Organization
The muscle fiber is structurally integrated through a complex network of cytoskeletal and membrane-associated proteins that link the intracellular contractile apparatus to the sarcolemma and extracellular matrix, ensuring efficient force transmission and mechanical stability. Central to this system is dystrophin, which connects the actin cytoskeleton to transmembrane proteins within the sarcolemma, forming part of a larger complex that anchors the muscle fiber to surrounding connective tissue.
Additionally, costameres—specialized protein assemblies aligned with Z-discs—mechanically couple myofibrils to the sarcolemma, enabling the lateral transmission of force generated within sarcomeres. This ensures that contractile forces are not only propagated longitudinally along myofibrils but also distributed across the cell membrane and into the extracellular matrix.
This integrated structural framework plays a critical role in maintaining cellular integrity during contraction, preventing membrane damage under mechanical stress, and enabling efficient transmission of force to higher levels of organization, including fascicles and whole muscle.
Exam Question
Explain how cytoskeletal and membrane-associated proteins, including dystrophin and costameres, contribute to force transmission and mechanical stability in skeletal muscle fibers, and analyze the consequences of disruption of these systems.”
FASCICULAR ORHGANIZATION

“Skeletal Muscle sag hariadhi” – Hariadhi via Wikimedia Commons.
Licensed under CC BY-SA 4.0
AI Generated Illustiaton ( MyoAnatomy)
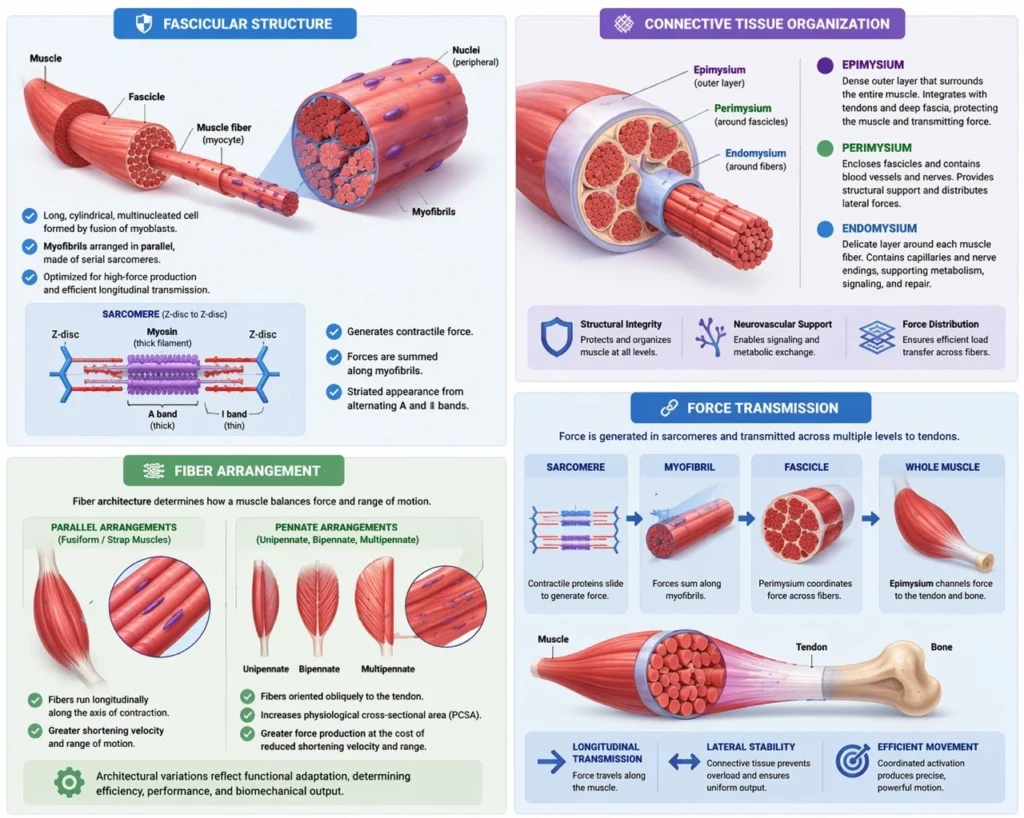
Fascicular Structure
Skeletal muscle fibers (myocytes) are organized into fascicles, which represent intermediate structural and functional units within the muscle. Each fascicle contains a coordinated assembly of muscle fibers aligned to optimize collective force generation. These bundles are enclosed by the perimysium, a dense connective tissue sheath that provides both mechanical support and structural compartmentalization.
The perimysium serves as a critical conduit for blood vessels, lymphatics, and nerves, ensuring adequate metabolic supply and synchronized neural activation of fibers within each fascicle.
Functionally, fascicles act as discrete yet integrated contractile units, allowing efficient coordination between individual muscle fibers while maintaining structural integrity under mechanical load. This hierarchical organization enhances both force precision and distribution, contributing to the overall functional versatility of skeletal muscle
Exam Question
Explain the structural organization of fascicles in skeletal muscle, including the role of the perimysium in vascular, neural, and mechanical integration, and discuss how this organization contributes to controlled and efficient muscle contraction.”
Connective Tissue Organization
Skeletal muscle is organized within a continuous, hierarchical connective tissue framework composed of the endomysium, perimysium, and epimysium, which together provide structural integrity, facilitate force transmission, and enable neurovascular integration across all levels of muscle organization.
The endomysium is a delicate layer of connective tissue that surrounds individual muscle fibers. It contains a fine network of collagen fibers, capillaries, and nerve endings, creating a microenvironment that supports metabolic exchange and electrical signaling at the cellular level. Importantly, the endomysium plays a key role in lateral force transmission, allowing forces generated by individual fibers to be distributed to adjacent fibers and the surrounding matrix.
The perimysium encloses groups of muscle fibers into fascicles and represents a critical intermediate structural layer. It contains larger blood vessels and nerves and provides a scaffold for coordinated activation of fiber groups. Mechanically, the perimysium is essential for distributing contractile forces across fibers within a fascicle, reducing localized stress and enhancing overall mechanical efficiency.
The epimysium is the outermost dense connective tissue layer that surrounds the entire muscle. It integrates with tendons and deep fascia, enabling the transmission of force generated within the muscle to the skeletal system. The epimysium ensures global structural integrity, protects the muscle from external mechanical stress, and serves as the final conduit through which force is transferred to produce movement.
Together, these layers form a continuous mechanical network that enables both longitudinal and lateral force transmission, ensuring that microscopic contractile activity is effectively translated into coordinated macroscopic movement. This system also maintains structural stability during repeated contraction and prevents mechanical damage under high loads.
Exam Question
Describe the hierarchical organization of connective tissue in skeletal muscle (endomysium, perimysium, epimysium), and analyze how each layer contributes to force transmission, structural integrity, and coordinated muscle function.”
Fiber Arrangment
Within each fascicle, muscle fibers are arranged according to specific architectural patterns that determine the functional properties of the muscle, particularly the balance between force generation and range of motion.
In parallel arrangements (fusiform or strap muscles), fibers run longitudinally along the axis of contraction. This configuration allows for greater shortening velocity and range of motion, as the fiber length directly contributes to excursion.
In contrast, pennate arrangements (unipennate, bipennate, multipennate) feature fibers oriented obliquely relative to the tendon. This increases the physiological cross-sectional area (PCSA), enabling greater force production despite shorter fiber length. However, this comes at the cost of reduced contraction velocity and range.
These architectural variations reflect functional adaptation, allowing muscles to specialize based on mechanical demands. The arrangement of fibers within fascicles therefore represents a critical determinant of muscle performance, efficiency, and biomechanical output.
Exam Question
Compare parallel and pennate muscle fiber arrangements in terms of structural organization, physiological cross-sectional area, and functional outcomes, and explain how these architectural differences influence force generation and range of motion.”
Force Transmission
Force generated within individual sarcomeres is transmitted through a multilevel mechanical continuum that integrates intracellular and extracellular structures. At the cellular level, force propagates longitudinally along myofibrils and is simultaneously distributed laterally through the cytoskeleton and connective tissue interfaces.
Within the fascicle, the perimysium coordinates force distribution among fibers, preventing localized overload and ensuring uniform mechanical output. The surrounding connective tissue network (endomysium → perimysium → epimysium) enables efficient transmission of force from individual muscle fibers to the tendon, allowing microscopic contractile activity to be translated into macroscopic movement.
This system ensures mechanical stability, load redistribution, and coordinated activation, optimizing both efficiency and durability during repeated contraction.
Exam Question
“Describe the mechanisms of longitudinal and lateral force transmission in skeletal muscle, and explain how connective tissue organization contributes to efficient and coordinated muscle function.”
