Bone Remodeling
Bone remodeling is a tightly regulated, continuous biological process by which bone tissue undergoes renewal, structural adaptation, and metabolic regulation throughout life. It is driven by the coordinated activity of bone-resorbing osteoclasts and bone-forming osteoblasts, which replace aged or microdamaged bone with newly synthesized mineralized matrix.
This dynamic coupling ensures the maintenance of skeletal strength, preservation of mineral (calcium–phosphate) homeostasis, and optimization of bone architecture in response to mechanical loading. Through this process, bone continuously adapts to functional demands while preventing the accumulation of structural defects.
In contrast to bone modeling, which shapes the skeleton during growth via uncoupled formation and resorption, bone remodeling persists throughout life and is essential for maintaining the microstructural integrity of mature bone tissue.
In adult humans, approximately 5–10% of the total skeletal mass is remodeled annually, reflecting a tightly controlled turnover of bone matrix necessary for long-term structural maintenance and physiological function.
BASIS
AI -Generated illustration-MyoAantomy
Overview
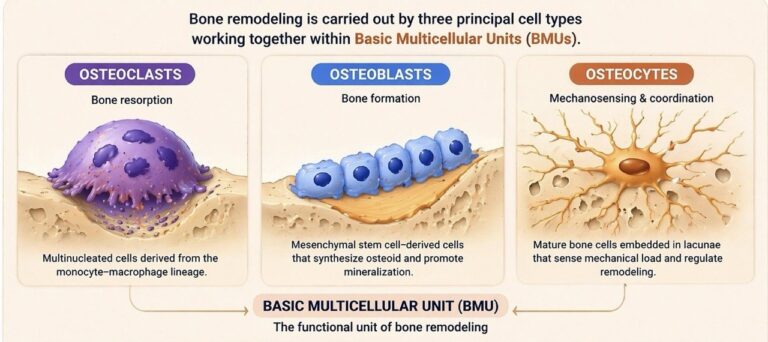
Bone remodeling is mediated by the coordinated activity of 3 principal bone cell types:
osteoclasts, osteoblasts, and osteocytes, which operate within specialized anatomical units known as- basic multicellular units (BMUs).
Osteoclasts
Osteoclasts are large, multinucleated cells derived from the monocyte–macrophage lineage (hematopoietic origin), specialized for bone resorption.
Their differentiation and activation are tightly regulated by the RANK–RANKL–OPG signaling axis, where:
RANKL (expressed by osteoblasts and osteocytes) binds to RANK receptors on osteoclast precursors
This interaction drives osteoclast differentiation, activation, and survival
Activated osteoclasts adhere to the bone surface and form a sealed microenvironment known as the resorption lacuna (Howship’s lacuna).
Within this compartment, osteoclasts mediate bone degradation via:
Hydrochloric acid (H⁺ secretion) → dissolves inorganic hydroxyapatite
Proteolytic enzymes (e.g., cathepsin K) → degrade organic collagen matrix
This coordinated process: Results in the removal of aged, damaged, or mechanically compromised bone, enabling subsequent bone formation.
Exam Question
How does activation of the RANK–RANKL–OPG signaling axis regulate osteoclast differentiation and function, and by what precise cellular and biochemical mechanisms do activated osteoclasts degrade both the inorganic hydroxyapatite and organic collagen matrix within the resorption lacuna (Howship’s lacuna)?
Osteoblasts
Osteoblasts are bone-forming cells derived from mesenchymal stem cells, responsible for the synthesis and mineralization of bone matrix.
They produce osteoid, the organic component of bone, composed primarily of:
Type I collagen fibers
Non-collagenous proteins (osteocalcin, osteopontin, bone sialoprotein)
Following matrix deposition, osteoblasts initiate mineralization, during which:
Calcium and phosphate ions precipitate as hydroxyapatite crystals
This process confers mechanical strength and rigidity
Fate of osteoblasts:
Differentiate into osteocytes (embedded mechanosensors)
Become bone-lining cells
Undergo apoptosis
Exam Question
How do osteoblasts coordinate the synthesis of osteoid and its subsequent mineralization into hydroxyapatite, and how does their differentiation into osteocytes or bone-lining cells contribute to long-term maintenance of bone structure and mechanosensory regulation?
Osteocytes
Osteocytes are mature, terminally differentiated bone cells embedded within the mineralized bone matrix, residing in small cavities known as lacunae. They extend long cytoplasmic processes through microscopic channels called canaliculi, forming an extensive interconnected cellular network that facilitates intercellular communication and nutrient exchange.
Functionally, osteocytes serve as primary mechanosensors of bone, detecting mechanical strain, fluid shear stress, and microdamage within the matrix. In response to mechanical stimuli, they regulate bone remodeling through the secretion of signaling molecules, most notably sclerostin, which modulates osteoblast activity and bone formation.
Through this mechanotransductive capability, osteocytes play a central role in coordinating the balance between bone formation and resorption, directing remodeling toward regions subjected to increased mechanical demand, and thereby maintaining structural integrity and adaptive optimization of bone architecture.
Exam Question
How do osteocytes function as mechanosensors within the lacuno–canalicular network, and by what molecular mechanisms (e.g., sclerostin signaling) do they regulate osteoblast activity and direct site-specific bone remodeling in response to mechanical loading?
REMODELEING CYCLE
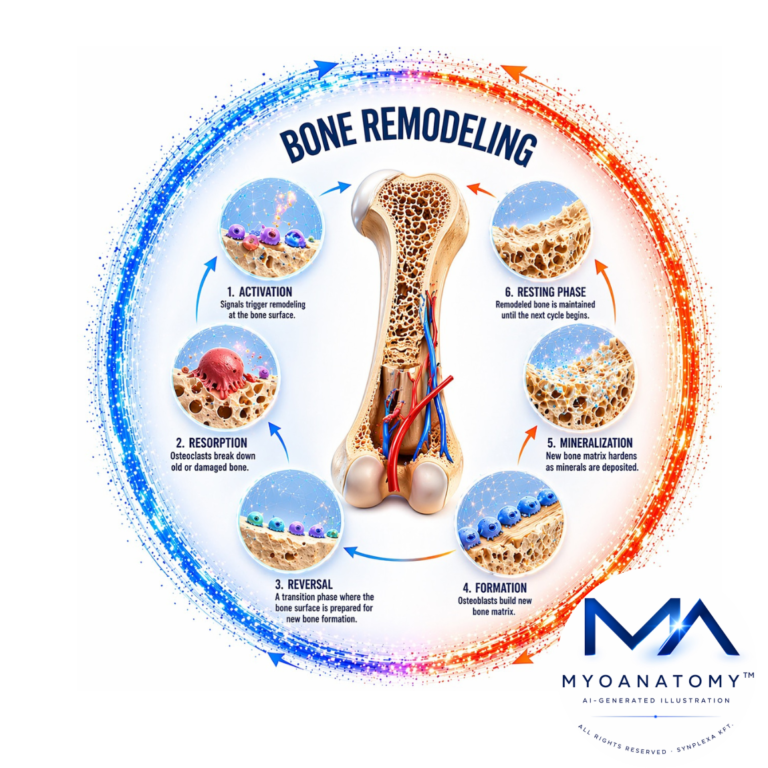
Activation Phase
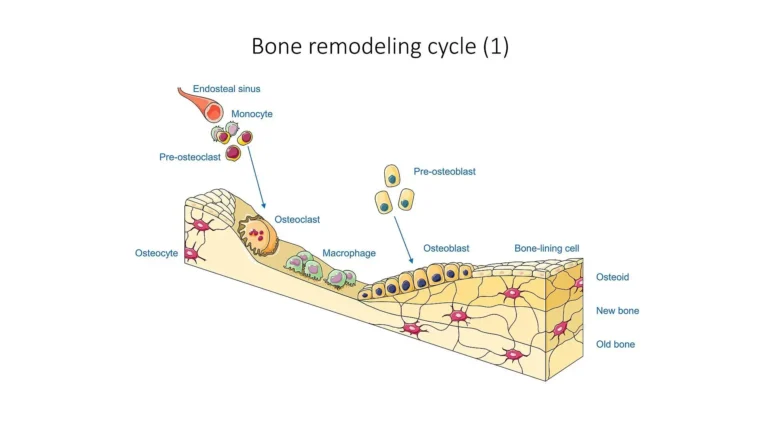
“Bone remodeling cycle” by SMART-Servier Medical Art (Laboratories Servier), via Wikimedia Commons Licensed under CC BY-SA3.0
The activation phase initiates bone remodeling through the recruitment and differentiation of osteoclast precursors. Mechanical loading, microdamage, and systemic hormonal signals (e.g., PTH) stimulate osteocytes and bone-lining cells to release key regulatory molecules, including RANKL, macrophage colony-stimulating factor (M-CSF), and pro-inflammatory cytokines.
RANKL binds to RANK receptors on osteoclast precursors, promoting their differentiation, fusion, and activation, while M-CSF supports precursor survival and proliferation. This tightly regulated signaling cascade establishes the cellular basis for targeted bone resorption at sites of structural demand.
Exam Question
How do osteocytes and bone-lining cells integrate mechanical and hormonal signals to regulate the RANK–RANKL–M-CSF axis, and how does this signaling cascade control osteoclast precursor recruitment, differentiation, and site-specific initiation of bone remodeling?
Resorption Phase
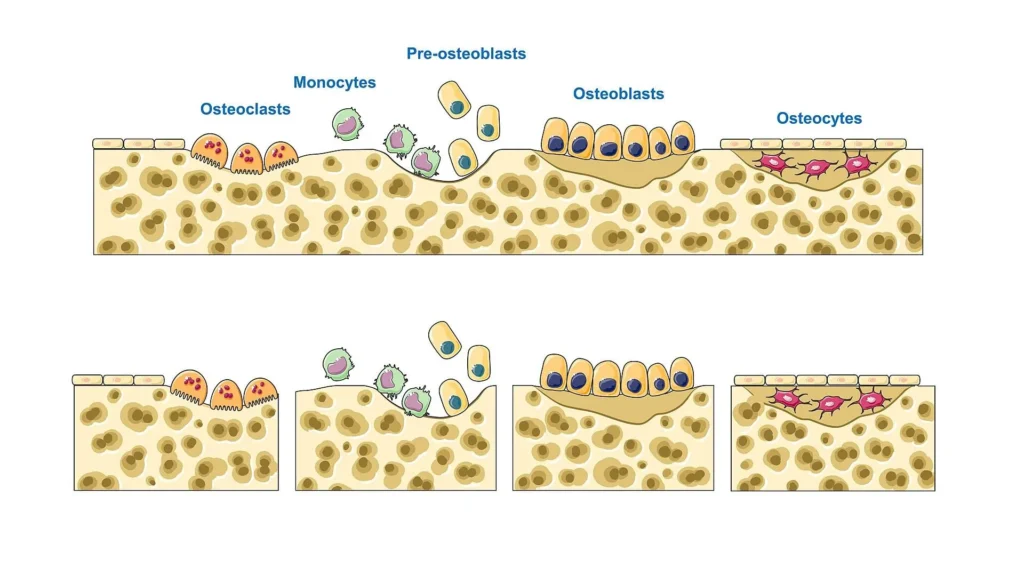
“Bone remodeling cycle” by SMART-Servier Medical Art (Laboratories Servier), via Wikimedia Commons Licensed under CC BY-SA3.0
During the resorption phase, activated osteoclasts adhere to the bone surface and establish a specialized sealed microenvironment known as the resorption lacuna (Howship’s lacuna). Within this compartment, osteoclasts generate a ruffled border to increase surface area for secretion.
They actively secrete hydrogen ions (via proton pumps) to acidify the environment, dissolving the inorganic hydroxyapatite matrix, while releasing proteolytic enzymes (e.g., cathepsin K) to degrade the organic collagen matrix. This coordinated process results in the formation of a resorption pit and removal of structurally compromised bone.
Exam Question
By what cellular specializations and biochemical mechanisms do osteoclasts establish the resorption lacuna and achieve coordinated degradation of both mineral (hydroxyapatite) and organic (collagen) components of bone matrix?
Reversal Phase
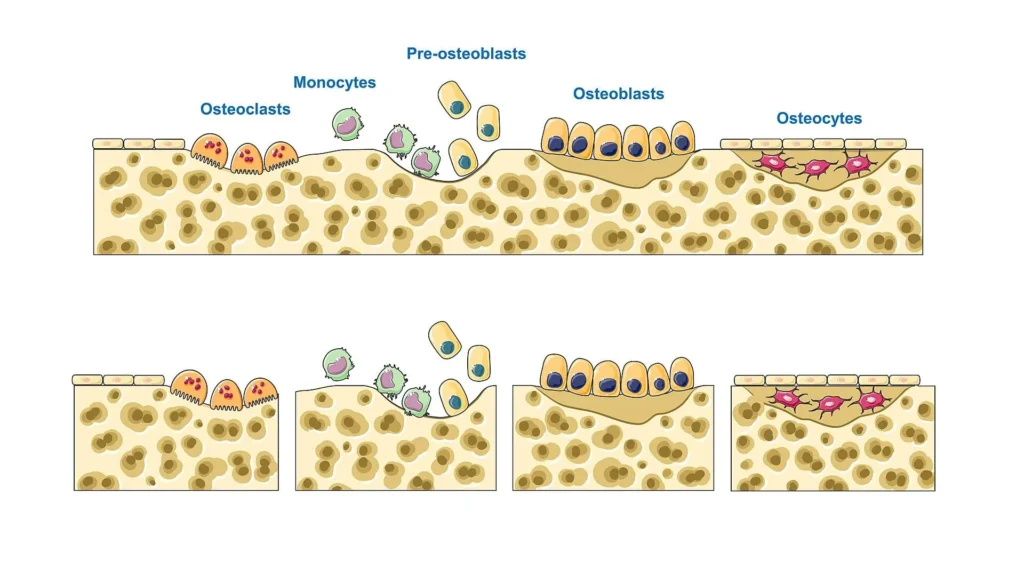
“Bone remodeling cycle” by SMART-Servier Medical Art (Laboratories Servier), via Wikimedia Commons Licensed under CC BY-SA3.0
The reversal phase represents a critical transitional stage between bone resorption and formation. Following osteoclast apoptosis or migration, mononuclear reversal cells colonize the resorption surface.
These cells remove residual debris, modify the microenvironment, and secrete signaling molecules that recruit and differentiate osteoblast precursors. This phase ensures proper coupling between resorption and formation, preventing structural discontinuity and enabling precise spatial coordination of new bone deposition.
Exam Question
How do reversal cells mediate the transition from osteoclastic resorption to osteoblastic formation, and what mechanisms ensure effective coupling between these phases to maintain structural continuity of bone?
Formation Phase

“Bone remodeling cycle” by SMART-Servier Medical Art (Laboratories Servier), via Wikimedia Commons Licensed under CC BY-SA3.0
During the formation phase, osteoblasts synthesize and deposit osteoid, the unmineralized organic matrix of bone composed primarily of type I collagen fibers and non-collagenous proteins.
Osteoid provides a structural scaffold for subsequent mineralization. Osteoblasts align along the resorption surface and coordinate matrix deposition in a highly regulated manner, restoring bone architecture. Over time, some osteoblasts differentiate into osteocytes, become bone-lining cells, or undergo apoptosis.
Exam Question
How do osteoblasts coordinate osteoid synthesis, spatial organization, and differentiation into osteocytes or lining cells, and how does this process restore bone architecture following resorption?
Mineralization Phase

“Bone remodeling cycle” by SMART-Servier Medical Art (Laboratories Servier), via Wikimedia Commons Licensed under CC BY-SA3.0
The mineralization phase involves the deposition of inorganic minerals within the osteoid matrix. Calcium and phosphate ions precipitate to form hydroxyapatite crystals, which integrate with collagen fibers to confer mechanical strength and rigidity.
This process occurs in two stages: an initial rapid phase followed by a slower maturation phase that may continue for months. Proper mineralization is essential for restoring the functional load-bearing capacity of bone.
Exam Question
How does the process of osteoid mineralization proceed at the molecular level, including hydroxyapatite crystal formation, and how does this process restore the mechanical strength of bone over time?
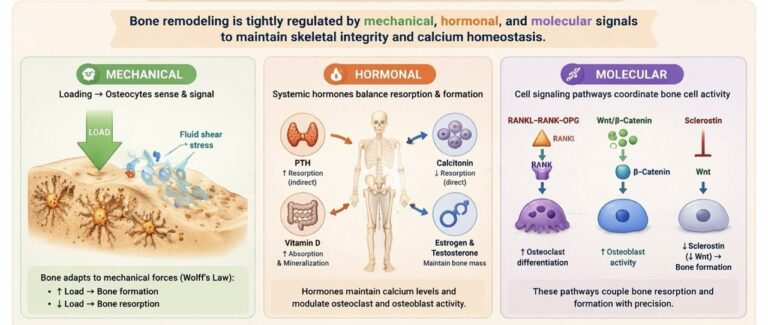
REGULATION
AI-Generated illustration-MyoAantomy
Mechanical
Bone remodeling is fundamentally regulated by mechanical loading, following Wolff’s law, whereby bone adapts its structure to the magnitude and direction of applied forces. Mechanical strain generates fluid shear stress within the lacuno–canalicular system, which is detected by osteocytes acting as mechanosensors.
This mechanotransduction process leads to downregulation of sclerostin and activation of osteoblast-mediated bone formation in regions of increased load. Conversely, reduced mechanical stimulation (e.g., immobilization) promotes osteoclast-mediated bone resorption, resulting in decreased bone mass.
Thus, mechanical forces dynamically regulate bone architecture, optimizing strength, density, and structural alignment according to functional demand.
Exam Question
How does mechanical loading translate into cellular responses within bone via osteocyte-mediated mechanotransduction, and how does this process regulate the balance between osteoblast activity and osteoclast-mediated resorption according to Wolff’s law?
Hormonal
Bone remodeling is tightly controlled by systemic hormonal regulation, which modulates the balance between bone formation and resorption.
Parathyroid hormone (PTH) increases serum calcium levels by indirectly stimulating osteoclast activity through osteoblast-mediated expression of RANKL.
Calcitonin antagonizes this effect by directly inhibiting osteoclast function.
Vitamin D (calcitriol) enhances intestinal calcium absorption and supports mineralization of bone matrix.
Hormones (estrogen and testosterone) play a critical role in maintaining bone mass by suppressing osteoclast activity and promoting osteoblast survival.
These hormonal systems act in coordination to maintain calcium homeostasis and skeletal integrity.
Exam Question
How do PTH, calcitonin, vitamin D, and sex hormones interact at the cellular level to regulate osteoclast and osteoblast activity, and how does this integrated hormonal control maintain calcium homeostasis and bone density?
Molecular
Bone remodeling is governed by highly coordinated molecular signaling pathways that regulate cellular differentiation and activity.
The RANK–RANKL–OPG system is central to osteoclastogenesis: RANKL promotes osteoclast differentiation, while osteoprotegerin (OPG) acts as a decoy receptor inhibiting this process.
The Wnt/β-catenin pathway is critical for osteoblast differentiation and bone formation.
Sclerostin, produced by osteocytes, inhibits Wnt signaling and thereby suppresses osteoblast activity.
These molecular pathways ensure precise coupling between bone resorption and formation, allowing adaptive remodeling in response to mechanical and metabolic demands.
Exam Question
How do the RANK–RANKL–OPG system, Wnt/β-catenin signaling, and sclerostin-mediated inhibition interact to coordinate osteoclastogenesis and osteoblast activity during bone remodeling? – Mineralization Phase
FUNCTIONAL ROLE
Mechanical Adaption
A central function of bone remodeling is adaptation to the mechanical environment. During normal physiological activity – such as weight-bearing, locomotion, and muscle contraction – bone is exposed to complex forces including compression, tension, bending, torsion, and shear.
Reduced loading
e.g., immobilization, bed rest, microgravity shifts remodeling toward bone resorption, leading to structural weakening.
Thus, remodeling ensures that bone architecture remains matched to functional demand.
Integration with the Musculoskeletal System
Bone does not function in isolation but as part of an integrated system with muscles, tendons, ligaments, and joints.
Muscle contractions generate traction forces at entheses → stimulate local bone reinforcement
Joint loading influences periarticular and subchondral bone structure. Remodeling therefore maintains structural compatibility between skeletal tissue and mechanical forces generated by movement.
Bone Integrity
Skeletal strength depends not only on bone mass but on material quality, including:
collagen organization
degree of mineralization
microarchitecture
Remodeling replaces older bone with organized lamellar bone, preserving the balance between:
stiffness (load-bearing capacity)
toughness (resistance to fracture)
Mineral Homeostasis
Bone remodeling is also supports the skeleton metabolic’s role as reservoir for calcium and phosphate.
Through controlled resorption and deposition, it contributes to:
maintenance of extracellular calcium levels
neuromuscular function
mineral balance for new bone formation
Healing Role
Following fracture repair, initial woven bone is mechanically weak and disorganized. Remodeling gradually replaces it with lamellar bone, restoring:
normal architecture
internal alignment
biomechanical strength
CLINICAL RELEVANCE
Osteoporosis
One of the most common disorders of bone remodeling is osteoporosis, which results from an imbalance favoring osteoclastic bone resorption over osteoblastic bone formation. The progressive loss of bone mass and deterioration of trabecular microarchitecture reduce the ability of bone to resist mechanical forces.
From a musculoskeletal perspective, osteoporosis leads to:
increased susceptibility to fragility fractures, particularly of the vertebrae, proximal femur, and distal radius
vertebral compression fractures, which alter spinal biomechanics and may lead to kyphotic deformity
impaired load distribution across joints, increasing the risk of secondary degenerative changes.
These structural alterations compromise the functional stability of the skeleton and significantly impair mobility and quality of life.
Paget Disease
Paget disease of bone represents another disorder of remodeling characterized by excessive and disorganized bone turnover. In this condition, hyperactive osteoclastic resorption is followed by rapid but poorly coordinated osteoblastic bone formation.
Although the affected bones may appear enlarged and dense radiographically, the newly formed bone is structurally abnormal, mechanically weak, and prone to deformation.
Musculoskeletal consequences include:
bone deformities such as bowing of long bones
altered joint alignment leading to secondary osteoarthritis
increased risk of pathological fractures
compression of adjacent nerves due to skeletal enlargement.
Thus, despite increased bone mass, the structural integrity of bone is compromised due to abnormal remodeling.
Osteopetrosis
In contrast to osteoporosis, osteopetrosis results from defective osteoclast function, leading to impaired bone resorption and failure of normal remodeling.
As a result, bone becomes excessively dense but structurally abnormal and brittle.
From a musculoskeletal standpoint, this condition leads to:
reduced ability to repair microdamage within bone
increased susceptibility to fractures despite increased bone density
narrowing of medullary cavities, which may affect bone marrow function
impaired skeletal modeling during growth.
The condition highlights the fact that bone strength depends not only on bone mass but also on normal remodeling and microarchitectural organization.
Remodeling Impact
Bone remodeling maintains skeletal strength and adaptation to mechanical load. Its disruption leads to microfractures, trabecular deterioration, cortical thinning, and impaired load distribution, weakening joint support and increasing fracture risk.
Clinically, it is essential for fracture healing, implant stability, and bone adaptation to physical stress. It also enables microdamage repair and structural maintenance, ensuring long-term skeletal stability and musculoskeletal function.
