Bone Architecture
Bone architecture refers to the structural organization of bone tissue at both the macroscopic (gross anatomical) and microscopic (histological) levels.
This hierarchical organization enables bone to fulfill its primary roles within the musculoskeletal system, including mechanical support, protection of vital organs, facilitation of movement, and efficient transmission and distribution of mechanical forces generated during locomotion and muscular activity.
The architecture of bone reflects a fundamental principle of biological design: optimization of strength with minimal mass, allowing skeletal structures to withstand mechanical stress while maintaining lightweight efficiency and functional adaptability.
GROSS STRCUTURE
AI -Generated Illustration-MyoAantomy
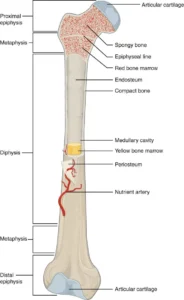
“Anatomy of Long Bone” by OpenStax College, via Wikimedia Commons. Licensed under CC BY SA 3.0
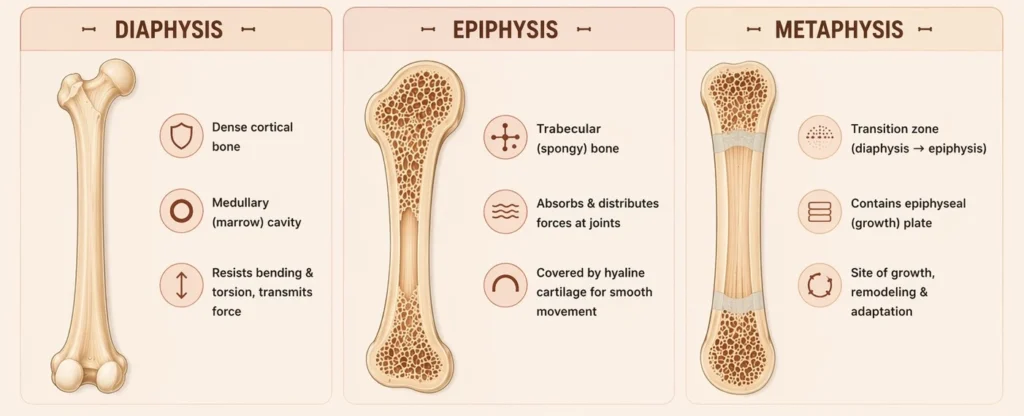
Diaphysis
The diaphysis represents the elongated central shaft of a long bone, composed predominantly of compact (cortical) bone, which forms a dense outer shell providing high mechanical strength and resistance to loading.
It encloses the medullary (marrow) cavity, which in adults primarily contains yellow bone marrow (adipose tissue) and contributes to weight reduction without compromising structural integrity.
The cylindrical geometry of the diaphysis is biomechanically optimized to resist bending and torsional stresses, enabling efficient force transmission along the longitudinal axis while maintaining a favorable strength-to-weight ratio.
Exam Question
How does the structural composition and cylindrical geometry of the diaphysis enable efficient resistance to bending and torsional stresses while facilitating longitudinal force transmission in long bones?
Epiphysis
The epiphyses are the expanded proximal and distal ends of long bones, composed mainly of trabecular (spongy) bone, organized as a lattice-like network of trabeculae.
This architecture allows effective absorption and distribution of forces transmitted across synovial joints, reducing localized stress.
The articular surfaces are covered by hyaline cartilage, which minimizes friction and facilitates smooth, low-resistance joint movement.
Exam Question
How does the trabecular architecture and articular cartilage of the epiphysis support force absorption, load distribution, and low-friction movement at synovial joints?
Metaphysis
The metaphysis is the transitional region between the diaphysis and epiphysis, characterized by a gradual shift from dense cortical bone to trabecular architecture.
During skeletal development, it contains the epiphyseal (growth) plate, a zone of hyaline cartilage responsible for longitudinal bone growth through endochondral ossification.
This region is highly metabolically active and structurally specialized to facilitate rapid bone remodeling and adaptation to mechanical stress, while maintaining structural continuity between the shaft and the articular ends.
Following skeletal maturity, the growth plate undergoes ossification, forming the epiphyseal line, marking the cessation of longitudinal growth.
Exam Question
How does the structural and biological specialization of the metaphysis, particularly its role as a transition zone containing the epiphyseal growth plate, support longitudinal bone growth, remodeling activity, and integration of mechanical forces between the diaphysis and epiphysis?
TISSUE TYPES
AI -Generated Illustration-MyoAantomy
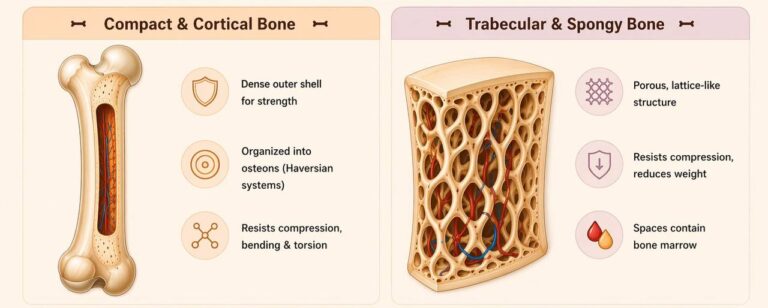
Compact & Cortical Bone
Compact (cortical) bone forms the dense outer shell of bone, providing the primary mechanical strength and rigidity of the skeleton, particularly in the diaphysis of long bones.
Microscopically, it is organized into osteons (Haversian systems), composed of concentric lamellae surrounding a central Haversian canal containing blood vessels and nerves.
Adjacent osteons are interconnected via Volkmann’s canals, enabling vascular and neural integration throughout the bone.
This highly organized structure allows compact bone to effectively resist compressive, bending, and torsional stresses, making it essential for load-bearing and force transmission during movement.
Exam Question
How does the osteonal organization of compact bone, including Haversian and Volkmann’s canal systems, enable efficient resistance to multidirectional mechanical stresses and support vascular integration within dense bone tissue?
Trabecular & Spongy Bone
Trabecular (spongy) bone consists of a porous, lattice-like network of trabeculae, arranged along lines of mechanical stress, optimizing structural efficiency.
This architecture enables bone to resist compressive forces while minimizing overall skeletal mass, contributing to a favorable strength-to-weight ratio.
The intertrabecular spaces contain bone marrow:
Red marrow → hematopoiesis
Yellow marrow → fat storage (adipose tissue)
Trabecular bone is predominantly found in regions subjected to multidirectional loading, such as the epiphyses of long bones, vertebrae, pelvis, and ribs, where it facilitates force distribution and shock absorption.
Exam Question
How does the trabecular architecture, aligned along lines of mechanical stress, allow spongy bone to achieve efficient load distribution, shock absorption, and reduction of skeletal mass while maintaining structural integrity?
BONE COVERING
AI-Generated Illustration-MyoAantomy
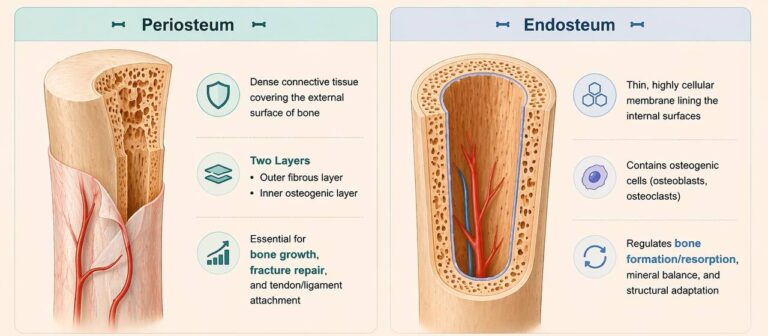
Periosteum
The periosteum is a dense connective tissue membrane covering the external surface of bone, except at articular cartilage–covered regions.
It consists of two distinct layers:
an outer fibrous layer, composed of collagen fibers, providing mechanical protection and structural support
an inner osteogenic layer, containing osteoprogenitor cells capable of differentiating into osteoblasts
The periosteum plays a critical role in bone growth (appositional growth), fracture repair, and serves as a site for attachment of tendons and ligaments via Sharpey’s fibers, enabling efficient force transmission between muscle and bone.
Exam Question
How does the bilaminar structure of the periosteum, including its fibrous and osteogenic layers, enable coordinated mechanical force transmission, appositional bone growth, and fracture repair, and what is the functional significance of Sharpey’s fibers in this context?
Endoosteum
The endosteum is a thin, highly cellular connective tissue lining the internal surfaces of bone, including the medullary cavity, trabecular surfaces, and Haversian canals.
It contains osteogenic cells, including osteoblasts and osteoclasts, and is actively involved in bone remodeling, regulating bone formation and resorption.
Functionally, the endosteum plays a central role in metabolic regulation of bone tissue, maintaining mineral homeostasis and enabling continuous structural adaptation to mechanical and physiological demands.
Exam Question
How does the cellular composition of the endosteum regulate the dynamic balance between bone formation and resorption, and how does this contribute to continuous structural adaptation and mineral homeostasis within the skeletal system?
MATRIX CELLS
AI-Generated Illustration-MyoAantomy
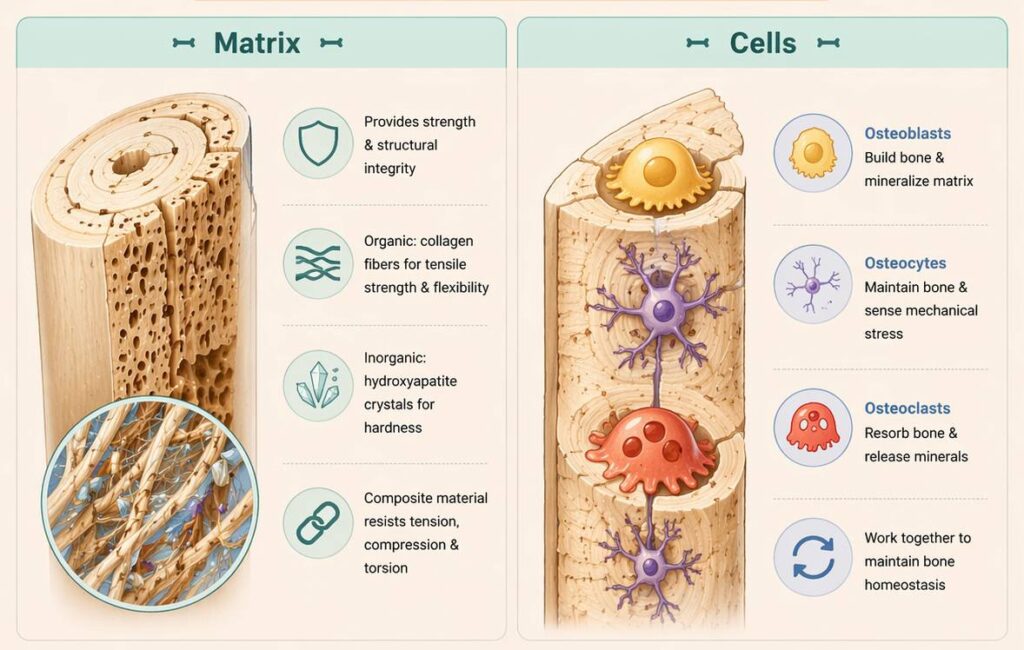
Matrix
Bone tissue consists of a mineralized extracellular matrix that provides both mechanical strength and structural integrity, together with specialized bone cells.
The organic component (osteoid) is composed predominantly of type I collagen fibers, which confer tensile strength and flexibility, allowing bone to resist stretching and bending forces.
The inorganic component consists primarily of hydroxyapatite crystals (calcium phosphate), which impart hardness and resistance to compressive forces.
The integration of these components creates a composite material optimized for simultaneous resistance to tension, compression, and torsion, enabling efficient load-bearing and force transmission within the musculoskeletal system.
Exam Question
How does the composite structure of bone matrix, integrating organic collagen fibers and inorganic hydroxyapatite crystals, enable simultaneous resistance to tensile, compressive, and torsional forces, and how does this contribute to overall skeletal biomechanics?
Cells
Bone metabolism is regulated by three principal cell types:
Osteoblasts → bone-forming cells responsible for the synthesis and mineralization of osteoid (bone matrix)
Osteocytes → mature bone cells embedded within the matrix, functioning as mechanosensors that regulate bone remodeling in response to mechanical stress
Osteoclasts → large, multinucleated cells derived from the monocyte lineage, responsible for bone resorption through enzymatic degradation and acidification
These cells operate in a coordinated manner to maintain bone homeostasis, enabling continuous remodeling, repair, and adaptation to mechanical loading.
Exam Question
How do osteoblasts, osteocytes, and osteoclasts function as a coordinated cellular system to regulate bone remodeling, and how does this interaction enable bone to adapt dynamically to mechanical stress and metabolic demands?
FUNCTIONAL ROLE
Structural Optimization
Bone architecture represents a hierarchically organized system that balances strength, stiffness, and weight.
Cortical bone provides resistance to bending and torsional forces, while trabecular bone distributes compressive loads, ensuring mechanical efficiency with minimal mass.
Structural Optimitization
Bone architecture represents a hierarchically organized system that balances strength, stiffness, and weight.
Cortical bone provides resistance to bending and torsional forces, while trabecular bone distributes compressive loads, ensuring mechanical efficiency with minimal mass.
Mechanoadaption
Through osteocyte-mediated mechanotransduction, bone continuously senses and responds to mechanical stimuli.
The interaction between cortical, trabecular, and periosteal components drives adaptive remodeling, preserving biomechanical integrity under varying loads.
Functional Integration
Bone architecture underpins skeletal support, joint stability, and coordinated force transmission during multiaxial movement, enabling efficient interaction between muscles, joints, and supporting structures.
CLINICAL RELEVANCE
Fracture Susceptibility
Disruption of trabecular architecture and reduction in bone density, as seen in osteoporosis, impair load distribution and significantly increase the risk of fragility fractures.
Diagnostic Assessment
Evaluation of cortical thickness and trabecular alignment is fundamental in imaging and clinical diagnostics, providing insight into bone strength, fracture risk, and underlying pathology.
Therapeutic Modulation
Bone remodeling capacity forms the basis of treatment strategies, where controlled mechanical loading and pharmacological interventions promote structural recovery and restore mechanical competence.
Pathological Alterations
Conditions such as stress fractures, subchondral degeneration, and metastatic disease highlight the relationship between altered mechanical loading, disrupted architecture, and adverse clinical outcomes.
