Venous Drainage
Arterial supply is a hierarchically organized vascular network delivering oxygen, metabolic substrates, and signaling molecules to tissues. It originates from the left ventricle → aorta → elastic arteries → muscular arteries → arterioles → capillaries, forming a system that enables both high-volume flow and precise regional regulation.
In the musculoskeletal system, arterial perfusion sustains the metabolic activity and structural integrity of muscle, bone, joints, and connective tissues. At the cellular level, it supports oxidative phosphorylation (ATP production), protein synthesis, collagen turnover, and bone remodeling—processes essential for contraction, repair, and adaptation to mechanical load.
Perfusion is dynamically regulated via local metabolic signals (hypoxia, ↑CO₂, ↓pH), endothelial factors (NO), and autonomic control, allowing rapid matching of blood flow to demand – especially in skeletal muscle, where energy requirements increase dramatically during activity.
Beyond nutrient delivery, arterial circulation contributes to thermoregulation, metabolic waste clearance, and maintenance of tissue homeostasis.
AI-generated illustration ( MyoAnatomy)
Large Vein, Medium Vein, and Venule (Histological Structure)” – OpenStax College via Wikimedia Commons.
Licensed under CC BY 3.0
Description
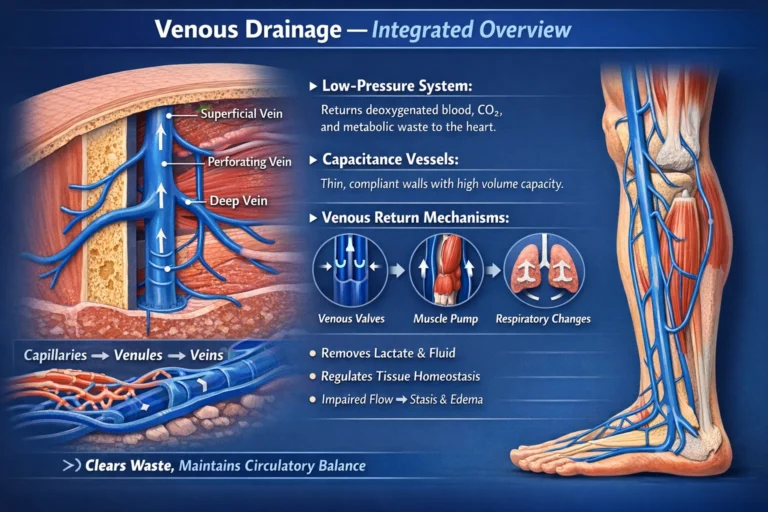
The venous system is organized as a hierarchical network of vessels that progressively collect blood from peripheral tissues and return it to the heart. Within the musculoskeletal system, this venous network plays an essential role in removing metabolic waste products generated by muscle contraction, regulating tissue fluid balance, and maintaining efficient circulatory dynamics.
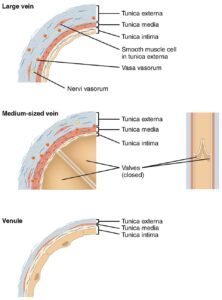
Venous vessels differ structurally from arteries in several important ways. Their walls are thinner and more compliant, and they contain a relatively larger lumen, allowing veins to function as capacitance vessels capable of storing significant volumes of blood. Despite these structural differences, veins retain the same fundamental histological layers as arteries:
tunica intima – an endothelial lining that facilitates smooth blood flow
tunica media – a layer containing smooth muscle and elastic fibers
tunica adventitia – an outer connective tissue layer that provides structural support and anchors the vessel to surrounding tissues.
As blood flows away from capillary beds, venous vessels increase progressively in size, forming a hierarchical sequence that includes venules, medium-sized veins, and large veins.
Venules
Venules represent the smallest components of the venous circulation and receive blood directly from capillary networks within tissues. These vessels collect deoxygenated blood and initiate its transport toward larger veins.
Structurally, venules possess extremely thin walls composed primarily of endothelial cells and a minimal connective tissue layer. In many regions, the tunica media is poorly developed or absent. Because of their thin walls and relatively high permeability, venules remain active sites of fluid exchange and leukocyte migration, playing an important role in inflammatory and immune responses.
Within musculoskeletal tissues, venules form dense networks surrounding muscle fibers, bone trabeculae, and synovial membranes, allowing efficient removal of metabolic by-products generated during tissue activity.
Medium-Sized Veins
Multiple venules converge to form medium-sized veins, which serve as the primary conduits for blood returning from local tissue regions. These veins possess a more developed vascular wall structure than venules.
The tunica media of medium-sized veins contains a modest layer of smooth muscle cells and elastic fibers that provide limited contractile ability and structural support. The tunica adventitia, composed of collagenous connective tissue, is typically the thickest layer and anchors the vein within surrounding tissues.
A defining feature of many medium-sized veins – particularly those of the limbs -is the presence of venous valves, which are folds of the tunica intima reinforced by connective tissue. These valves prevent retrograde blood flow and are critical for maintaining unidirectional circulation toward the heart, especially in regions where blood must travel against gravitational forces.
In the musculoskeletal system, medium-sized veins frequently accompany arteries and peripheral nerves, forming neurovascular bundles that supply skeletal muscles, bones, and joints. This anatomical association allows coordinated regulation of blood supply and drainage within functional musculoskeletal compartments.
Llarge Veins
Large Veins
Large veins represent the final collecting vessels of the venous circulation, receiving blood from multiple smaller tributaries and transporting it toward the heart.
Examples of major large veins include:
femoral vein, draining the lower limb; subclavian vein, draining the upper limb ; internal jugular vein, draining the head and neck.
Structurally, large veins possess a relatively thin tunica media but a well-developed tunica adventitia containing abundant collagen fibers and longitudinal smooth muscle bundles. This structural arrangement provides strength and flexibility while allowing these vessels to accommodate substantial volumes of blood.
Large veins ultimately converge into the superior vena cava and inferior vena cava, which deliver venous blood directly to the right atrium of the heart, completing the systemic venous return.
AI-genrated illustration ( MyoAnatomy)
Description
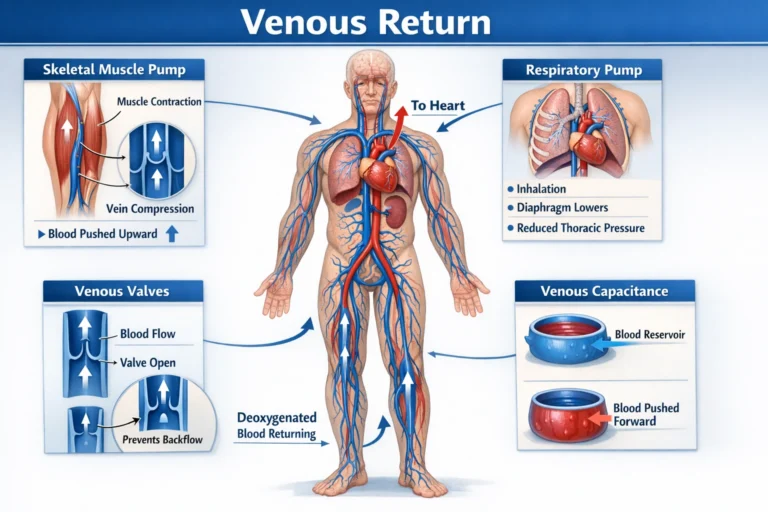
Venous return refers to the transport of deoxygenated blood from peripheral tissues back to the heart through the venous circulation. Because venous blood flows under relatively low intravascular pressure, several physiological mechanisms assist in propelling blood toward the central circulation. These mechanisms are particularly important in the limbs, where venous blood must frequently travel against gravitational forces.
The efficiency of venous return is maintained through the coordinated action of skeletal muscle contraction, respiratory pressure changes, venous valves, and the capacitance properties of veins. Together, these mechanisms ensure that venous blood from musculoskeletal tissues is returned efficiently to the heart, thereby sustaining circulatory homeostasis.
Skeletal Muscle Pump
The skeletal muscle pump represents one of the most important mechanisms facilitating venous return from the limbs. Deep veins are located within muscular compartments and are therefore subject to compression during muscular contraction.
When skeletal muscles contract, they exert pressure on adjacent veins, compressing the vessel walls and propelling blood proximally toward the heart. The presence of venous valves ensures that this movement occurs in a unidirectional manner, preventing retrograde flow when the muscles relax.
During rhythmic muscular activity – such as walking, running, or repetitive limb movements – this pumping action significantly enhances venous return. In the lower limbs, the muscle pump of the calf muscles (particularly the gastrocnemius and soleus) is especially important, often referred to as the “peripheral heart” due to its major role in maintaining venous circulation.
Prolonged immobility, such as during long periods of sitting or bed rest, can impair the skeletal muscle pump and lead to venous stasis, increasing the risk of deep vein thrombosis (DVT).
Respiratory Pump
Venous return is also facilitated by pressure changes occurring during respiration, a mechanism known as the respiratory pump.
During inspiration, contraction of the diaphragm increases the volume of the thoracic cavity, producing negative intrathoracic pressure. This pressure reduction draws venous blood from the abdominal and peripheral veins toward the thoracic veins and the right atrium.
Simultaneously, the descent of the diaphragm increases intra-abdominal pressure, compressing abdominal veins and further promoting the upward movement of venous blood toward the heart.
During expiration, intrathoracic pressure rises slightly; however, venous valves prevent backward flow of blood, ensuring continued movement toward the central circulation.
Through this cyclical process, respiration assists in maintaining continuous venous return, particularly from the abdominal and lower limb venous systems.
Venous Capacitance
Veins function as capacitance vessels, meaning they are capable of holding large volumes of blood due to their thin walls and high compliance. At any given time, a substantial proportion of the body’s total blood volume is contained within the venous system.
The capacity of veins to store and mobilize blood plays an important role in regulating venous return and overall circulatory dynamics. Changes in venous tone, mediated by smooth muscle within the venous wall and influenced by autonomic nervous system activity, can alter venous capacity and affect blood flow toward the heart.
Sympathetic stimulation causes venoconstriction, which reduces venous capacity and pushes blood toward the central circulation, thereby increasing venous return and cardiac preload. Conversely, venous dilation allows veins to store greater volumes of blood, contributing to the regulation of systemic blood distribution.
This dynamic regulation of venous capacitance is essential during physiological states such as exercise, hemorrhage, and postural changes, where rapid adjustments in circulating blood volume are required.
Venous Return
The mechanisms of venous return operate in a coordinated manner to ensure efficient circulation despite the relatively low pressure within the venous system. The skeletal muscle pump, respiratory pump, and venous capacitance collectively facilitate the movement of blood from peripheral tissues back to the heart.
Within the musculoskeletal system, these mechanisms are particularly important for maintaining adequate circulation during physical activity. As muscles contract, both the muscle pump and metabolic vasodilation enhance circulation, while respiratory movements and venous tone adjustments ensure that venous blood continues to flow efficiently toward the central circulation.
Metabolic Clearence
The venous system maintains the biochemical environment required for musculoskeletal function by removing carbon dioxide, lactate, hydrogen ions, and metabolic by-products generated during cellular activity.
This process preserves acid–base balance and transcapillary exchange gradients, preventing intracellular acidosis that would otherwise impair calcium handling and actin–myosin cross-bridge cycling. Efficient venous outflow is therefore essential for sustaining cellular metabolism, contractile function, and tissue viability, particularly under conditions of increased metabolic demand.
Muscle Performance
During muscular contraction, rapid accumulation of metabolites threatens excitation–contraction coupling and reduces force-generating capacity. The venous system, augmented by the skeletal muscle pump, facilitates continuous clearance of these substances while maintaining pressure gradients necessary for arterial inflow. This preserves neuromuscular efficiency, delays fatigue onset, and sustains force production and endurance. Thus, venous return is directly coupled to the ability of muscle to generate and maintain mechanical output.
Fluid Dynamics
Venous circulation plays a central role in regulating microvascular pressure, interstitial fluid balance, and capillary exchange efficiency.
As capacitance vessels, veins modulate blood volume distribution and maintain the pressure gradient between arterial inflow and venous outflow, which is essential for effective tissue perfusion. Impaired venous return elevates hydrostatic pressure, disrupts Starling forces, and leads to edema, reduced oxygen diffusion, and compromised nutrient delivery, ultimately impairing tissue function and structural integrity.
Tissue Repair
The venous system is critical for tissue repair by clearing inflammatory mediators, cellular debris, and catabolic by-products, thereby facilitating an environment conducive to regeneration.
It also contributes to thermoregulation through redistribution of heat generated during muscular activity. By maintaining circulatory continuity and supporting efficient recovery, venous drainage ensures restoration of tissue function, adaptation to mechanical load, and prevention of chronic degeneration.
Venous Stasis
Venous pathology commonly arises from disruption of normal flow dynamics, particularly under conditions of reduced mobility, endothelial injury, or hypercoagulability (Virchow’s triad). Because venous circulation operates under low pressure and depends on the skeletal muscle pump and valve competence, impaired flow leads to venous stasis, promoting thrombus formation.
In deep vein thrombosis (DVT), intraluminal clot obstructs venous return, elevating venous pressure and impairing tissue drainage. This results in edema, pain, and reduced perfusion efficiency, while posing a systemic risk of pulmonary embolism, highlighting the critical link between local venous dysfunction and life-threatening complications.
Chronic Venous Insufficiency
Chronic venous insufficiency (CVI) results from valvular incompetence, leading to retrograde blood flow and sustained venous hypertension. Elevated venous pressure disrupts microcirculatory exchange, increasing capillary hydrostatic pressure and promoting fluid extravasation into interstitial tissues.
Over time, this produces chronic edema, tissue hypoxia, and inflammatory changes, impairing nutrient delivery and waste removal. Clinically, CVI manifests as skin changes, ulceration, and reduced muscular efficiency, reflecting progressive deterioration of tissue integrity under conditions of persistent circulatory imbalance.
Compartment Syndrome
In compartment syndrome, increased intracompartmental pressure initially compromises venous outflow, leading to rapid accumulation of interstitial fluid and further pressure elevation.
This creates a vicious cycle that ultimately impairs arterial inflow, resulting in global ischemia of muscles and nerves. The combined failure of venous drainage and arterial supply leads to cellular hypoxia, ATP depletion, and irreversible tissue damage if not promptly relieved. This condition underscores the interdependence of venous and arterial systems in maintaining musculoskeletal viability.
Clinical Integration
Venous dysfunction directly compromises metabolic clearance, fluid balance, and effective perfusion, transforming normal tissue mechanics into a state of metabolic constraint and structural stress. Impaired venous return leads to edema, reduced oxygen diffusion, delayed healing, and diminished muscle performance, ultimately limiting functional capacity.
In clinical practice – across orthopedics, vascular medicine, sports medicine, and rehabilitation – understanding venous anatomy and physiology is essential for diagnosing circulatory disorders, preventing complications, and optimizing recovery following musculoskeletal injury.
