Clinical Relevance
Human movement represents the integrated output of a neuromuscular–biomechanical continuum, in which muscle contraction, connective tissue transmission, joint mechanics, and neural control operate as a unified system. Skeletal muscle functions as the primary effector, but its output depends on coordinated interaction with tendons, joints, and neural pathways.
Force generated at the sarcomere level is transmitted through connective tissues to the skeleton, producing joint torque, while neural mechanisms regulate activation, timing, and feedback within a closed-loop control system.
Disruption at any level produces predictable biomechanical dysfunction – including impaired force, altered kinematics, and loss of coordination – typically resulting in compensatory adaptations and system-wide imbalance. Consequently, localized pathology often manifests as a global movement disorder, necessitating a systems-based clinical approach.
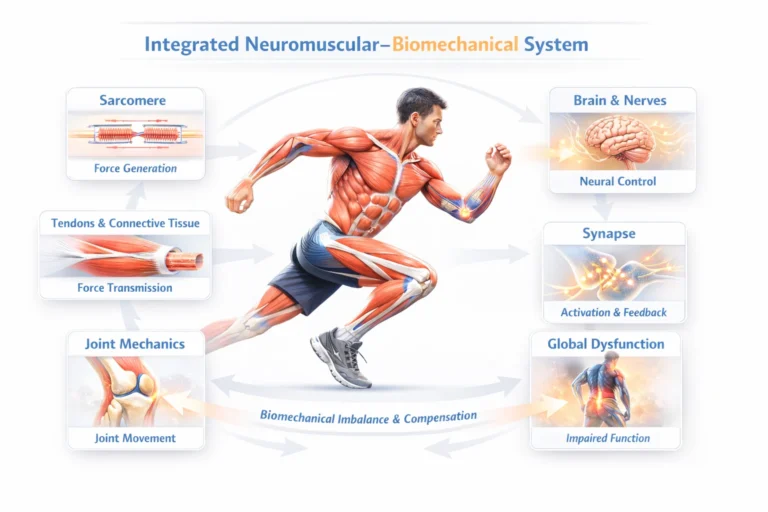
AI-generated illustration ( MyoAnatomy)
Impared Force Generation
Mechanism
Impaired force generation arises from structural and functional alterations within skeletal muscle, most notably reduction in physiological cross-sectional area (PCSA) and disruption of sarcomere integrity. These changes diminish the number of active cross-bridges available for force production, directly lowering maximal tension.
At a biomechanical level, this results in:
Altered force–length relationship → reduced force at optimal sarcomere length due to architectural disorganization
Impaired force–velocity relationship → decreased capacity for rapid force generation and power output
Reduced motor unit recruitment efficiency (in disuse or disease states)
Collectively, these impairments compromise both peak force and functional muscle performance across the range of motion.
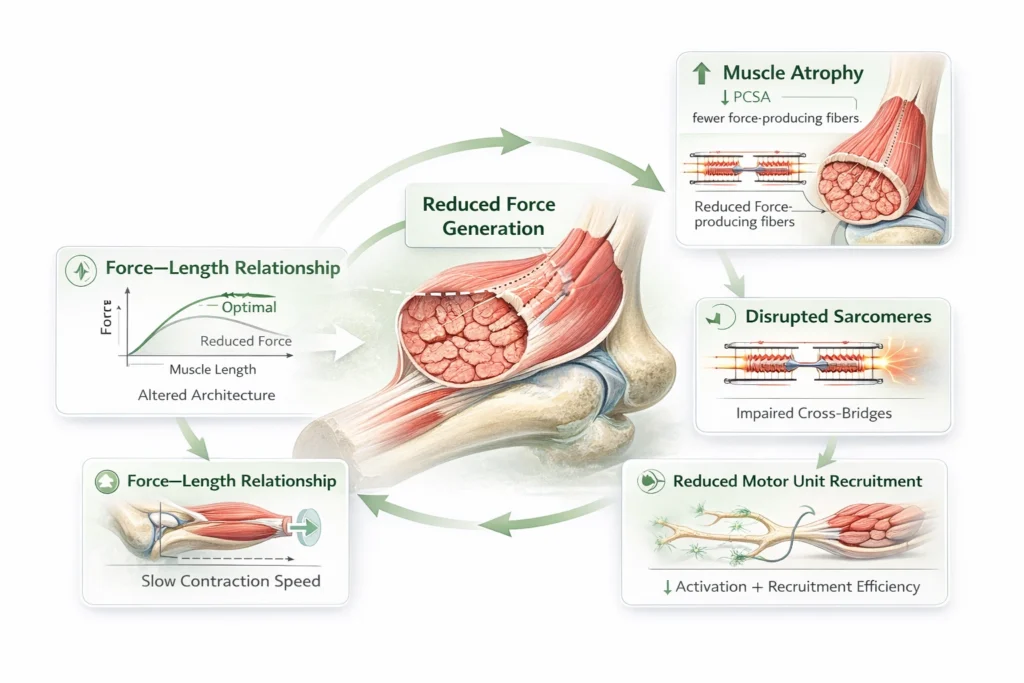
AI-generated illustration ( MyoAnatomy)
Functional Consequence
Reduced joint torque production, limiting effective movement
Loss of dynamic joint stabilization, increasing reliance on passive structures (ligaments, capsule)
Increased mechanical load on non-contractile tissues, predisposing to injury
Early onset of fatigue and diminished endurance capacity
Clinicoanalysis
Disuse atrophy → rapid muscle mass loss following immobilization or inactivity
Sarcopenia → age-related decline in muscle mass, strength, and neuromuscular efficiency
Myopathies → intrinsic muscle disorders affecting contractile proteins or metabolism
Clinical Insight- Muscle weakness represents not merely a reduction in strength, but a failure of force generation within a load-bearing system, leading to impaired joint control, altered biomechanics, and progressive structural stress.
In clinical practice, this manifests as:
Joint instability (e.g., quadriceps weakness → knee instability)
Compensatory muscle overuse
Increased risk of degenerative joint disease
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
Disrupted Neural Control
Mechanism
Efficient movement depends on precise central and peripheral neural regulation, integrating motor planning, execution, and feedback. This includes coordinated motor unit recruitment, rate coding, and synchronization, as well as continuous modulation via sensory (proprioceptive) input.
Neuromuscular pathology disrupts:
Neural drive to muscle → reduced or inappropriate activation
Temporal coordination → impaired sequencing and timing of muscle contraction
Sensorimotor integration → loss of accurate proprioceptive feedback
These disturbances impair the ability of the nervous system to generate smooth, adaptive, and task-specific motor output.
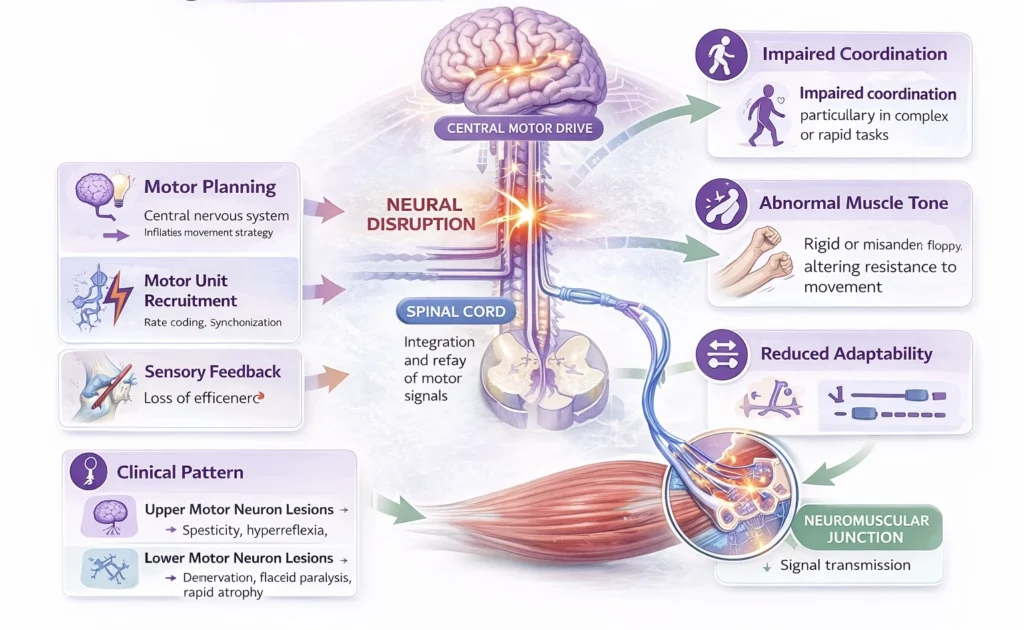
AI-generated illustration ( MyoAnatomy)
Functional Consequence
Impaired coordination and movement sequencing, particularly in complex or rapid tasks
Abnormal muscle tone (spasticity, rigidity, or flaccidity), altering resistance to movement
Loss of fine motor control and joint position sense
Reduced ability to adapt movement to changing mechanical demands
Clinicoanalysis
Upper motor neuron lesions → spasticity, hyperreflexia, stereotyped movement patterns
Lower motor neuron lesions → denervation, flaccid paralysis, rapid atrophy
Neuromuscular junction disorders → fatigable weakness with preserved initial strength
Clinical Insight (High-Yield)
Neuromuscular dysfunction transforms movement from a quantitatively reduced output into a qualitatively disordered process, characterized by impaired timing, abnormal coordination, and loss of adaptive control.
Clinically, this manifests as:
Abnormal synergy patterns and co-contraction
Inefficient, rigid, or poorly graded movements
Impared Force Transmission
Mechanism
Tendons and intramuscular connective tissues form a continuous mechanical interface that transmits force generated within muscle to the skeleton. Effective transmission depends on both structural integrity and viscoelastic properties of the collagen matrix.
Pathology disrupts:
Mechanical continuity → partial or complete rupture interrupts force transfer
Material properties → degeneration, reduced elasticity, and increased stiffness impair load transmission and energy storage
These alterations compromise the efficiency with which contractile force is converted into functional movement.
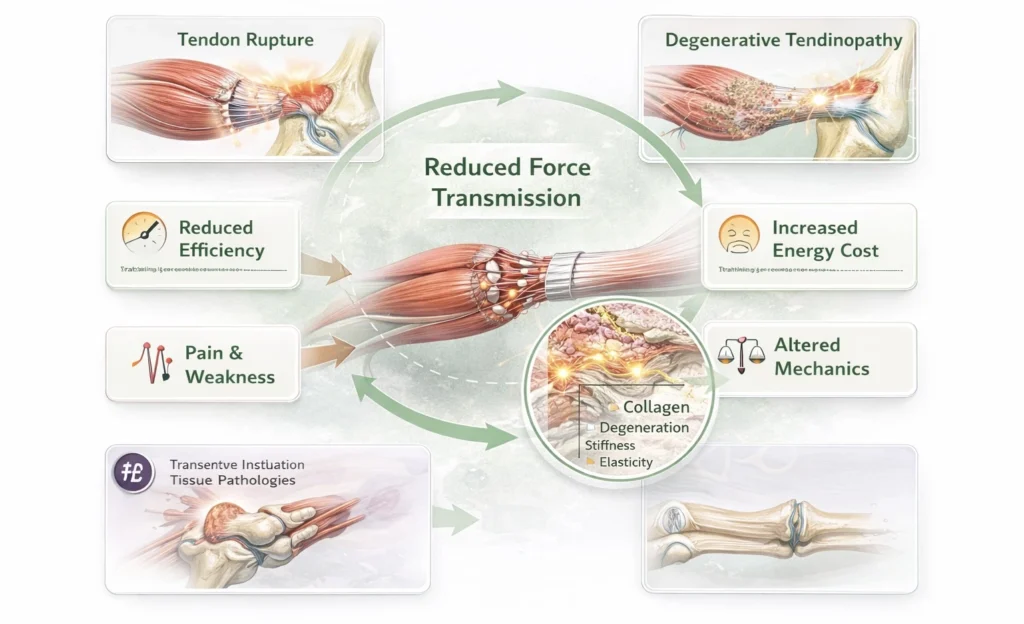
AI-generated illustration ( MyoAnatomy)
Functional Consequence
Functional Consequences
Reduced efficiency of force transfer, despite preserved muscle contraction
Increased energy expenditure during movement due to loss of elastic recoil
Pain-mediated inhibition of muscle activation, further reducing functional output
Decreased ability to generate smooth, coordinated force across joints
Biomechanical Impact
Altered moment arm mechanics, reducing effective joint torque
Impaired force–time characteristics, limiting rapid and explosive movements
Disruption of elastic energy storage and release, particularly critical in gait and locomotion
Clinicoanalysis
Tendinopathy → degenerative collagen disorganization with reduced tensile strength
Partial or complete tendon rupture → mechanical discontinuity and loss of function
Chronic overuse injuries → cumulative microtrauma with failed healing response
Clinical Insight (High-Yield)
Impaired force transmission represents a failure of the muscle–tendon unit as a mechanical continuum, in which intact muscle contraction cannot be effectively translated into movement.
Clinically, this manifests as:
Weakness disproportionate to muscle strength
Pain-associated movement limitation
Reduced mechanical efficiency and altered movement patterns
Altered Joint Mechanics
Mechanism
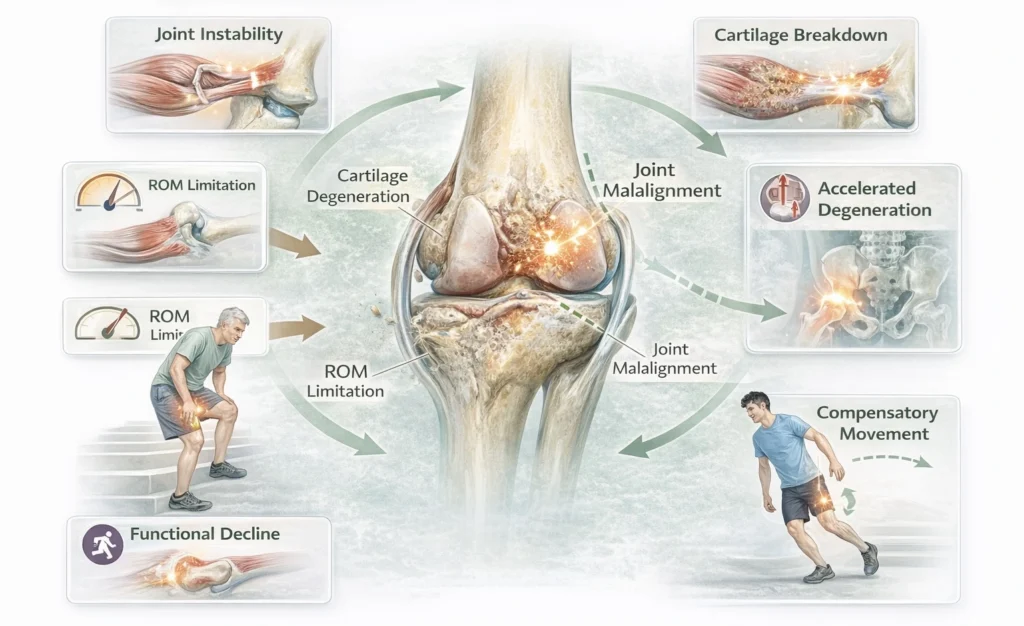
Joints establish the axis of rotation and mechanical framework within which muscle-generated forces are expressed. Normal function depends on optimal articular congruency, cartilage integrity, capsuloligamentous stability, and unrestricted range of motion.
Pathological processes disrupt:
Joint congruency → altered contact surfaces and uneven load distribution
Range of motion (ROM) → capsular restriction, structural limitation, or pain-mediated guarding
Load transmission → increased focal stress on cartilage, subchondral bone, and periarticular tissues
These changes modify joint kinematics and reduce the efficiency with which muscular force is translated into controlled movement.
AI-generated illustration ( MyoAnatomy)
Functional Consequence
Arthrogenic muscle inhibition, reducing activation of stabilizing muscles
Altered kinematics, including abnormal joint tracking and movement trajectories
Increased mechanical stress on adjacent joints and soft tissues
Reduced stability and efficiency during dynamic and load-bearing activities
Compensatory Adaptation
Redistribution of forces to adjacent joints or synergistic muscle groups
Development of maladaptive movement patterns, often reinforcing abnormal loading
Progressive biomechanical imbalance, contributing to secondary injury and degeneration
Clinicoanalysis
Osteoarthritis → cartilage degeneration, joint space narrowing, stiffness, reduced ROM
Inflammatory arthritis → synovial inflammation, pain, instability, functional limitation
Structural deformities → altered alignment, abnormal force vectors, chronic overload
Clinical Insight (High-Yield)
Joint pathology fundamentally alters the mechanical environment of movement, transforming normal force application into a compensatory and inefficient process.
Clinically, this results in:
Persistent pain with secondary muscular dysfunction
Asymmetric and energy-inefficient movement patterns
Progressive degeneration driven by abnormal load distribution
Impared Force Transmission
Mechanism
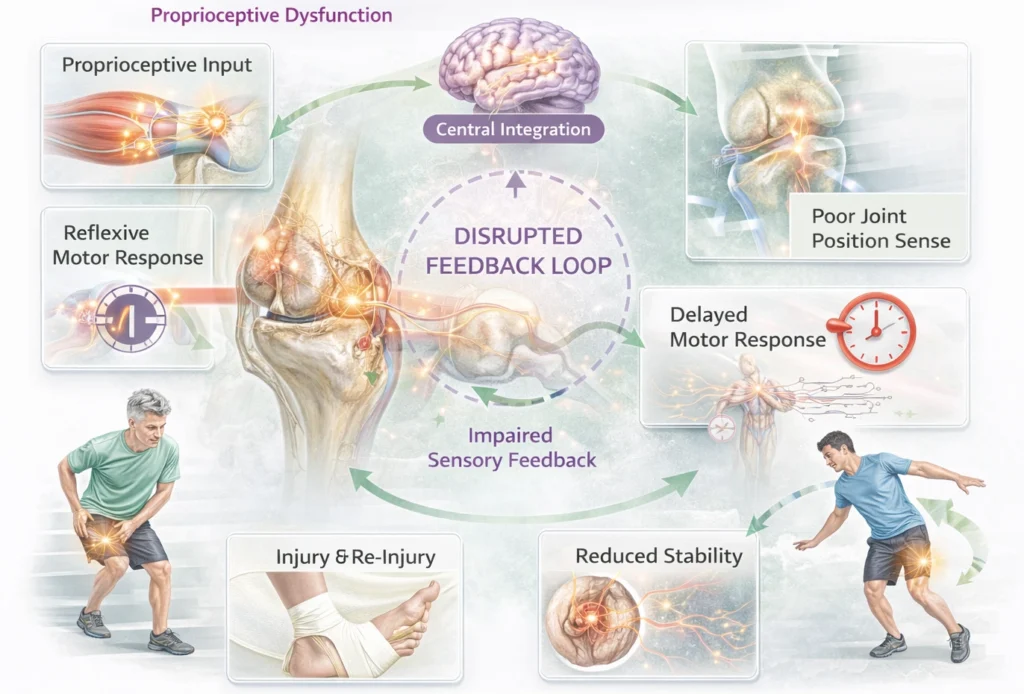
Effective movement depends on continuous integration of sensory input (proprioception, joint position, tension) with motor output. This feedback loop enables real-time adjustment of muscle activity.
Pathology disrupts:
Proprioceptive input from muscles, tendons, and joints
Central integration of sensory information
Reflexive motor responses
This leads to a breakdown in feedback-driven movement control.

AI-generated illustration ( MyoAnatomy)
Impared Sensiromotor
Mechanism
Effective movement depends on continuous integration of sensory input (proprioception, joint position, tension) with motor output. This feedback loop enables real-time adjustment of muscle activity.
Pathology disrupts:
Proprioceptive input from muscles, tendons, and joints
Central integration of sensory information
Reflexive motor responses
This leads to a breakdown in feedback-driven movement control.
AI-generated illustration ( MyoAnatomy)
Functional Consequence
Impaired joint position sense and kinesthesia
Delayed or inappropriate motor responses
Reduced stability, especially during dynamic or unpredictable tasks
Increased risk of injury and re-injury
Clinicoanalysis
Post-ligament injury (e.g., ACL) → loss of joint proprioception
Peripheral neuropathy → impaired sensory feedback
Central nervous system disorders affecting coordination
Clinical Insight
Movement dysfunction may arise not from weakness or structural damage, but from failure of sensory feedback integration, resulting in unstable and poorly controlled movement.
Clinically, this manifests as:
“Giving way” sensation despite adequate strength
Poor balance and coordination
Recurrent injuries due to impaired neuromuscular control
ROM Assessment
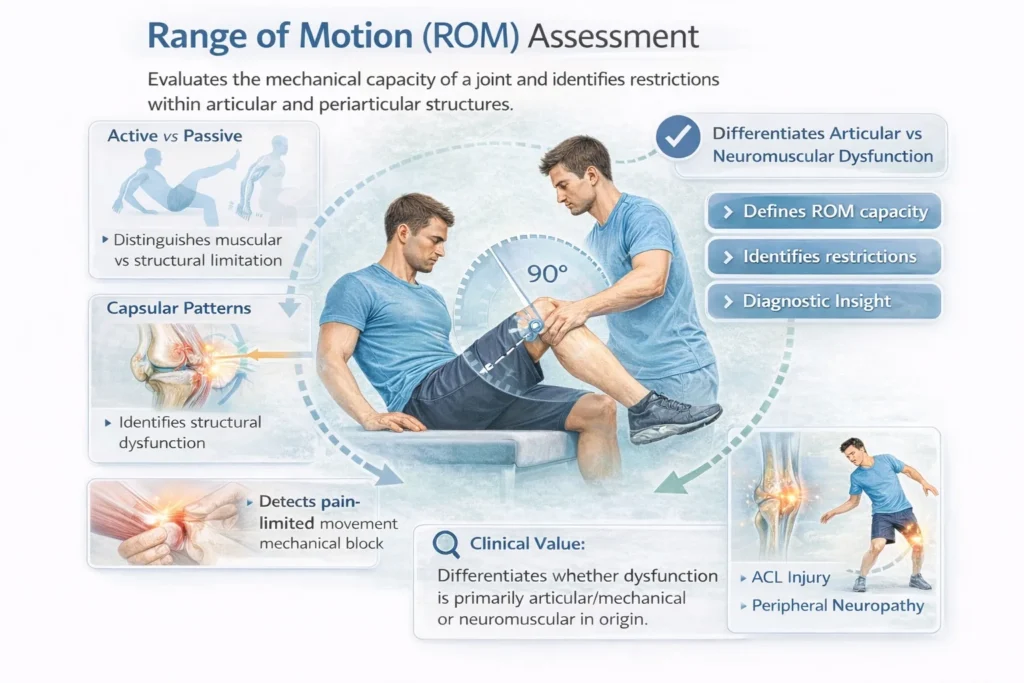
Range Of Motion ( ROM) evaluation defines the mechanical capacity of a joint and identifies restrictions within the articular and periarticular structures.
Differentiates active vs passive ROM, allowing distinction between muscular weakness and structural limitation
Identifies capsular patterns, ligamentous restriction, or joint incongruity
Detects pain-limited movement and mechanical block
Clinical value:
Provides insight into whether dysfunction is primarily articular/mechanical or neuromuscular in origin.
Manual Muscle Testing
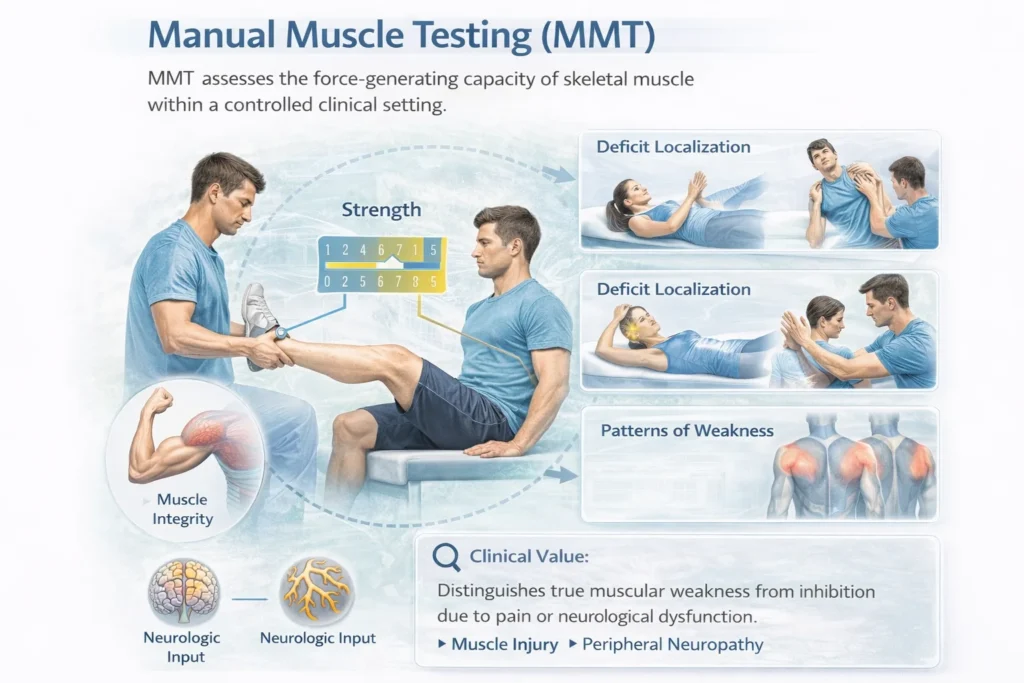
MMT assesses the force-generating capacity of skeletal muscle within a controlled clinical setting.
Evaluates muscle strength and functional integrity
Helps localize deficits to muscle tissue vs neural input
Identifies patterns of weakness across muscle groups
Clinical value:
Distinguishes true muscular weakness from inhibition due to pain or neurological dysfunction.
Gait analysis
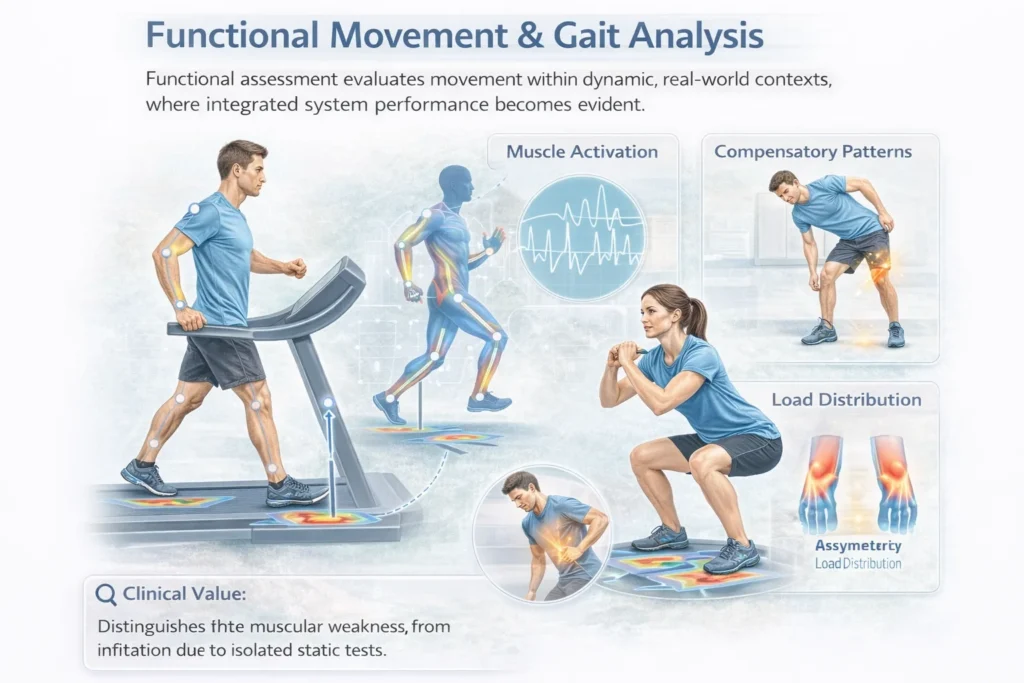
Functional assessment evaluates movement within dynamic, real-world contexts, where integrated system performance becomes evident.
Assesses coordination, timing, and sequencing of muscle activation
Identifies compensatory strategies, asymmetries, and inefficiencies
Evaluates load distribution during tasks such as walking, squatting, or reaching
Clinical value:
Reveals system-level dysfunction that may not be apparent in isolated static tests.
Neurological Assessment
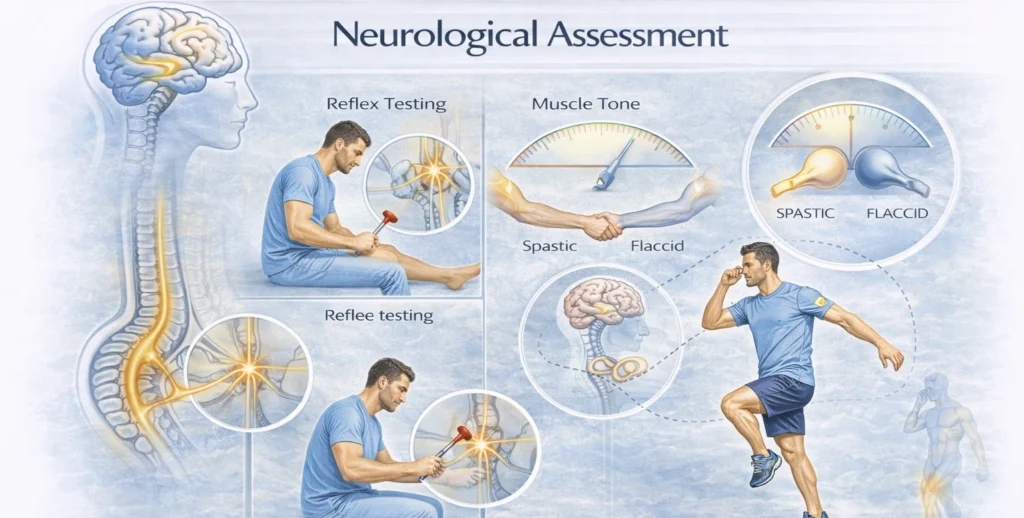
Neurological examination evaluates the integrity of central and peripheral control mechanisms governing movement.
Reflex testing → integrity of spinal pathways
Muscle tone → detection of spasticity or flaccidity
Coordination and proprioception → sensorimotor integration
Clinical value:
Identifies deficits in neural drive, motor control, and feedback regulation, essential for differentiating neurological from musculoskeletal pathology.
