Bone Ossification
Bone ossification is a tightly regulated process by which mesenchymal tissue is transformed into mineralized bone through coordinated cellular differentiation, osteoid synthesis, and hydroxyapatite deposition, producing a structurally competent and metabolically active skeletal framework. It underlies embryonic development, postnatal growth, fracture repair, and lifelong skeletal maintenance, ensuring mechanical stability, protection, and calcium–phosphate homeostasis.
This process is mediated by integrated activity of osteogenic lineage cells within a vascularized environment, where osteoblasts deposit a collagen-rich matrix that subsequently mineralizes, conferring rigidity and load-bearing capacity, while osteocytes coordinate adaptive responses and osteoclasts enable structural renewal.
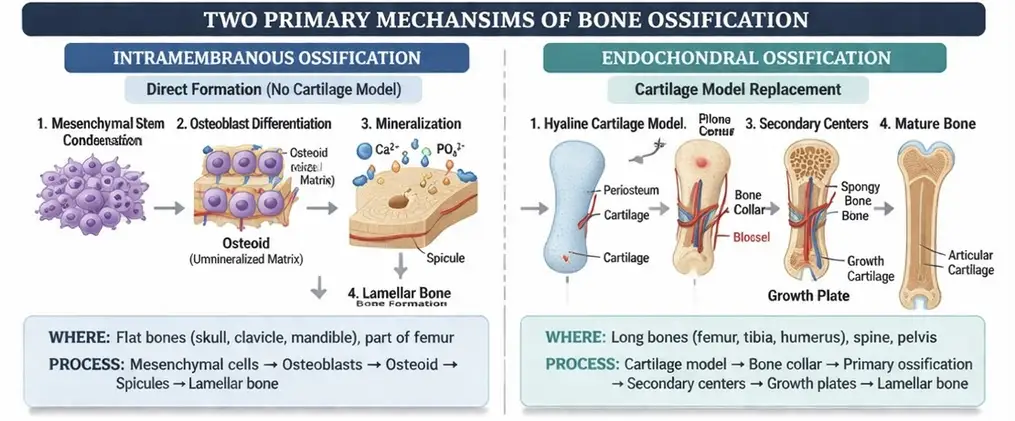
Ossification occurs via intramembranous and endochondral pathways, which differ in developmental sequence but converge in the formation of lamellar bone optimized for mechanical function. Throughout life, ossification remains dynamically regulated by mechanical forces, hormonal control, and molecular signaling, ensuring continuous adaptation, structural integrity, and metabolic balance of bone tissue.
AI-generated illustration (MyoAnatomy)
OSTEOGENESIS BASIS
AI -generated illustration (MyoAantomy)
Overview
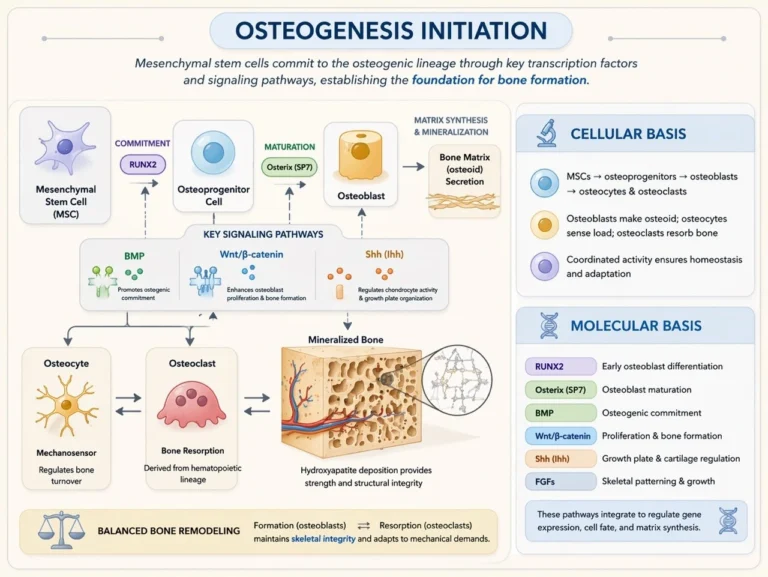
Bone formation is initiated by the differentiation of mesenchymal stem cells into osteogenic lineage cells. This differentiation is governed by several key transcription factors and signaling pathways that regulate osteoblast development and bone matrix synthesis.
Cellular Basis
Osteogenesis is initiated by the differentiation of mesenchymal stem cells (MSCs) into osteogenic lineage cells under tightly regulated developmental programs. These cells sequentially give rise to osteoprogenitors, osteoblasts, osteocytes, and osteoclasts, forming a coordinated cellular network responsible for bone formation and remodeling.
Osteoblasts synthesize osteoid and initiate mineralization, while embedded osteocytes function as mechanosensors, regulating bone turnover in response to mechanical and metabolic stimuli. Osteoclasts, derived from the hematopoietic lineage, mediate bone resorption, enabling continuous structural adaptation.
The dynamic balance between osteoblast-mediated formation and osteoclast-mediated resorption ensures skeletal integrity, functional adaptation, and maintenance of bone mass.
Exam Question
Explain the coordinated roles of osteoblasts, osteocytes, and osteoclasts in maintaining bone homeostasis, and describe how disruption of their balance leads to pathological bone remodeling.
Molecular Basis
Osteogenesis is regulated by tightly integrated transcription factors and signaling pathways that control osteogenic commitment, differentiation, and matrix production.
The transcription factor RUNX2 is essential for early osteoblast differentiation, while Osterix (SP7) drives osteoblast maturation. BMP signaling promotes osteogenic lineage commitment, and the Wnt/β-catenin pathway enhances osteoblast proliferation and bone formation.
Additionally, Indian hedgehog (Ihh) regulates chondrocyte activity and growth plate organization during endochondral ossification, while fibroblast growth factors (FGFs) contribute to skeletal patterning and longitudinal growth.
These pathways interact to precisely regulate gene expression, cellular differentiation, and matrix synthesis, ensuring coordinated bone formation and development.
Exam Question
Discuss the roles of RUNX2, Osterix, BMP, and Wnt/β-catenin signaling in osteoblast differentiation, and explain how their interaction coordinates bone formation during development and growth.
Initiation of Osteogeseisn
Bone formation is initiated by the commitment of mesenchymal stem cells (MSCs) to the osteogenic lineage, a process governed by tightly regulated transcriptional and signaling networks. This lineage specification is primarily driven by key transcription factors, including RUNX2 and Osterix (SP7), which direct osteoblast differentiation and maturation.
Simultaneously, signaling pathways such as BMP and Wnt/β-catenin promote osteogenic commitment, enhance osteoblast proliferation, and regulate bone matrix synthesis. These pathways coordinate gene expression patterns that control the transition from undifferentiated mesenchymal cells to fully functional osteoblasts capable of producing and mineralizing osteoid.
This initial phase establishes the cellular and molecular foundation for subsequent bone formation, ensuring precise regulation of skeletal development and structural integrity.
Exam Question
Explain the molecular mechanisms that govern mesenchymal stem cell commitment to the osteogenic lineage, including the roles of RUNX2, Osterix, BMP, and Wnt/β-catenin signaling in initiating bone formation..
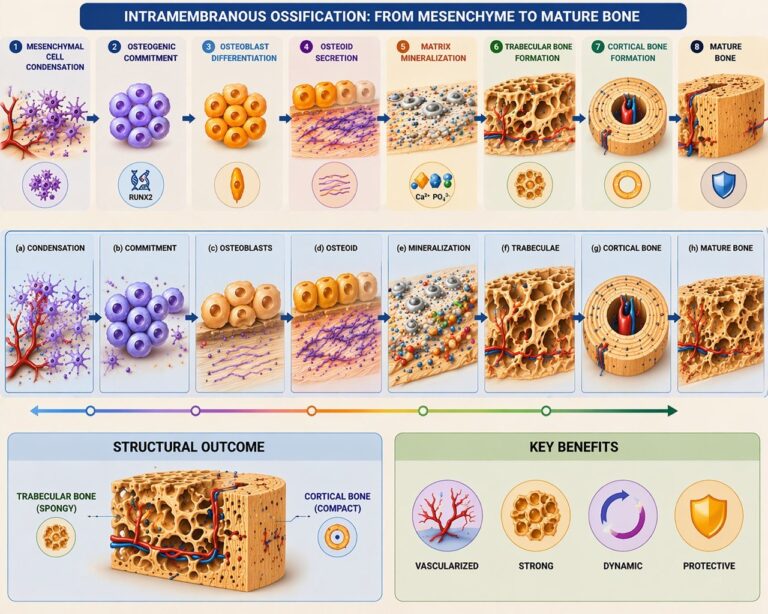
INTRAMEMBRANEOUS OSSIFICATION
AI -generated illustration (MyoAantomy)
Mesenchymal Cell Condensation
Mesenchymal stem cells aggregate within highly vascularized connective tissue, forming localized condensations that establish the initial sites of ossification. Within these microenvironments, cells undergo osteogenic commitment and differentiate into osteoprogenitor cells under the influence of transcription factors such as RUNX2. These progenitors subsequently give rise to osteoblasts, initiating the formation of primary ossification centers.
Osteoid Secretion
Differentiated osteoblasts synthesize and secrete osteoid, the unmineralized organic bone matrix composed predominantly of type I collagen fibers, along with proteoglycans and glycoproteins. This matrix provides a structural scaffold that determines the spatial organization of future mineral deposition and establishes the tensile framework of developing bone.
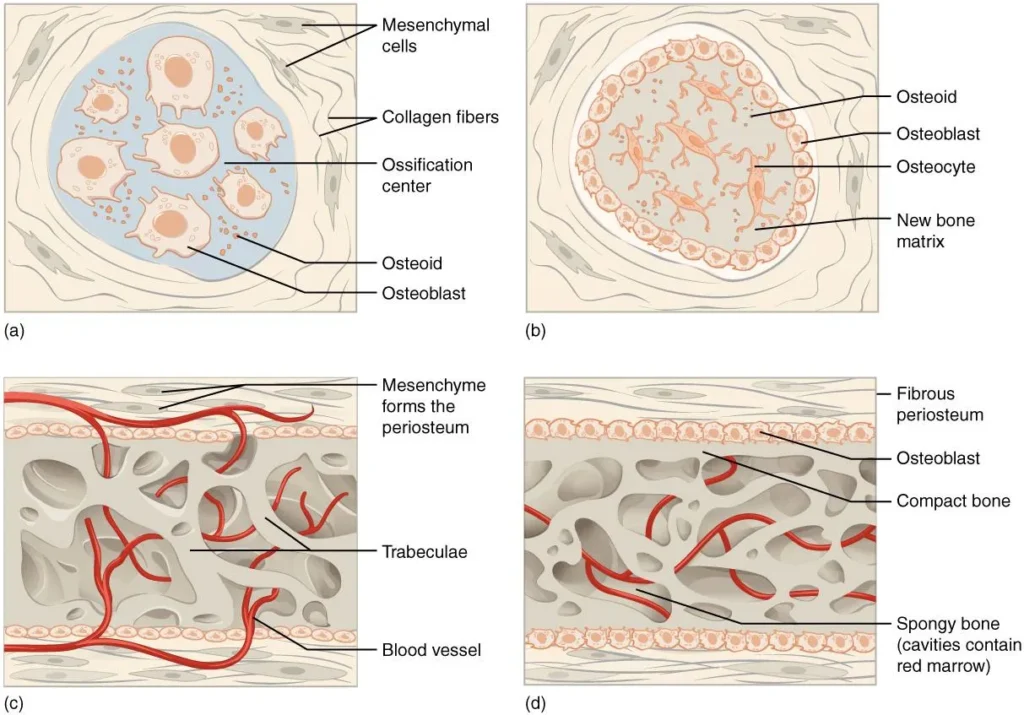
“Intramembraneous Ossification” by Open Stax College, from Anatomy&Physiology (OpenStax), via Wikimedia Commons. Licenswed under CC BY 3.0
Matrix Mineralization
Mineralization occurs as calcium (Ca²⁺) and phosphate (PO₄³⁻) ions accumulate within the osteoid matrix, leading to the formation of hydroxyapatite crystals. These crystals nucleate and propagate along collagen fibrils, converting the osteoid into rigid mineralized bone and conferring mechanical strength and load-bearing capacity.
Trabecular Bone Formation
Following mineralization, newly formed bone organizes into trabeculae, creating a three-dimensional network of spongy bone. This architecture is highly vascularized, facilitating nutrient exchange, cellular activity, and rapid bone turnover. The trabecular arrangement optimizes strength while minimizing mass, allowing efficient distribution of mechanical forces within developing bone.
Cortical Bone Formation
As ossification progresses, peripheral trabecular bone undergoes remodeling and compaction, forming dense cortical bone at the outer surfaces, while the internal region remains trabecular. This transition establishes a structural gradient, combining rigid external support with lightweight internal architecture, thereby enhancing mechanical strength and protecting underlying tissues
ENDOCHONDRAL OSSIFICATION
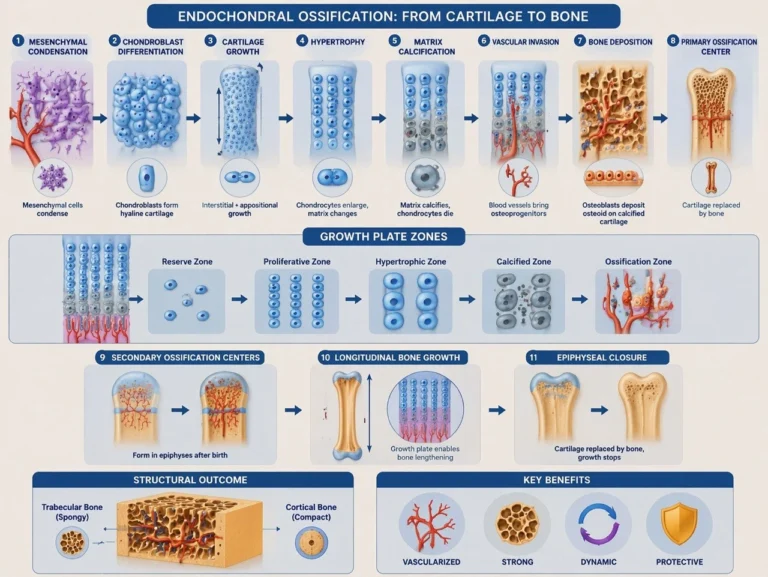
AI -generated illustration (MyoAantomy)
Development
Cartilage Model
Endochondral ossification is initiated by the differentiation of mesenchymal stem cells into chondroblasts, which synthesize a hyaline cartilage template that replicates the shape of the future bone. This cartilage model serves as a transient structural scaffold guiding subsequent bone formation.
Cartilage growth occurs via two complementary mechanisms:
Interstitial growth, driven by chondrocyte proliferation within the matrix, leading to internal expansion, and appositional growth, involving the addition of new cartilage layers at the surface from the perichondrium. Together, these processes enable controlled enlargement and morphological refinement of the developing skeletal element.
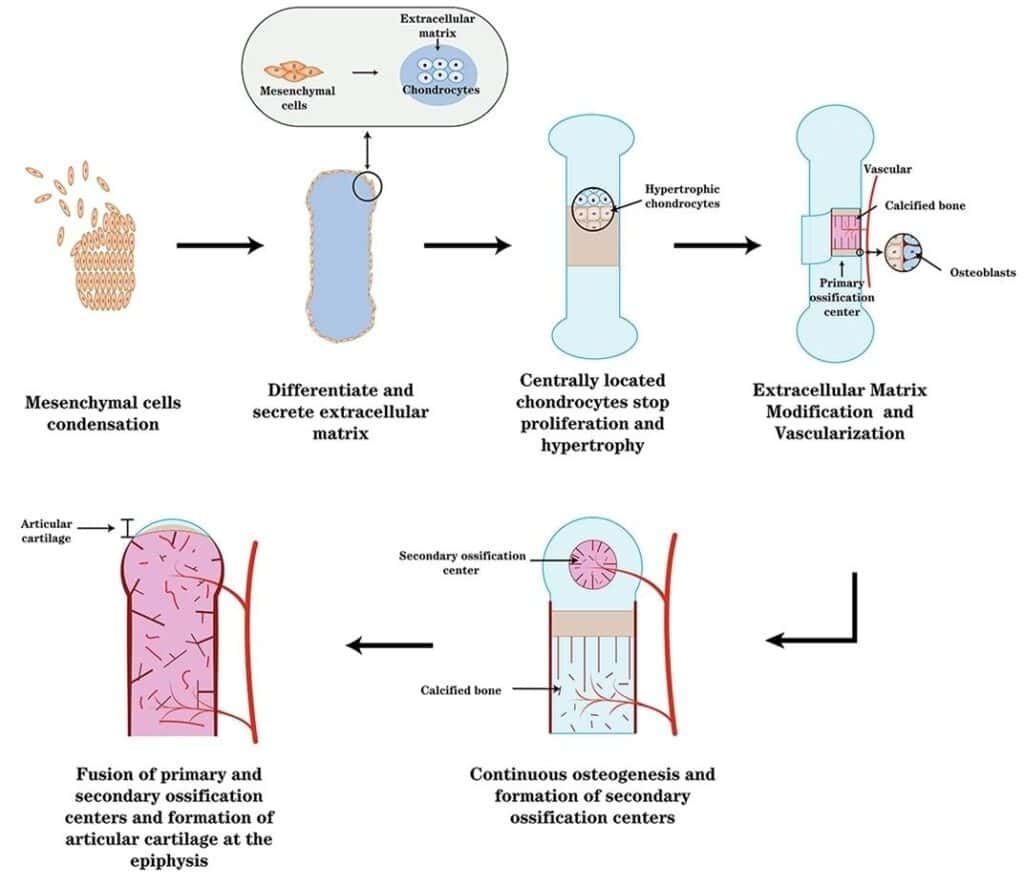
“Endochondral ossification and articular cartilage formation” by Zhangpeng Su et al., via NCBI/Wikimedia Commons. Licensed under CC BY SA 4.0
Formation Primary
Ossification Center
As the cartilage model enlarges, chondrocytes in the central diaphysis undergo hypertrophy, altering matrix composition and promoting calcification of the surrounding cartilage matrix. These hypertrophic chondrocytes secrete pro-angiogenic factors (e.g., VEGF), facilitating vascular invasion.
Penetration of blood vessels introduces osteoprogenitor cells and hematopoietic elements, transforming the perichondrium into a vascularized periosteum. Osteoprogenitor cells differentiate into osteoblasts, which begin depositing osteoid onto calcified cartilage remnants, initiating endochondral bone formation. This region becomes the primary ossification center, marking the transition from cartilage to bone and establishing the foundation for longitudinal growth and further skeletal development.strength and protecting underlying tissues.
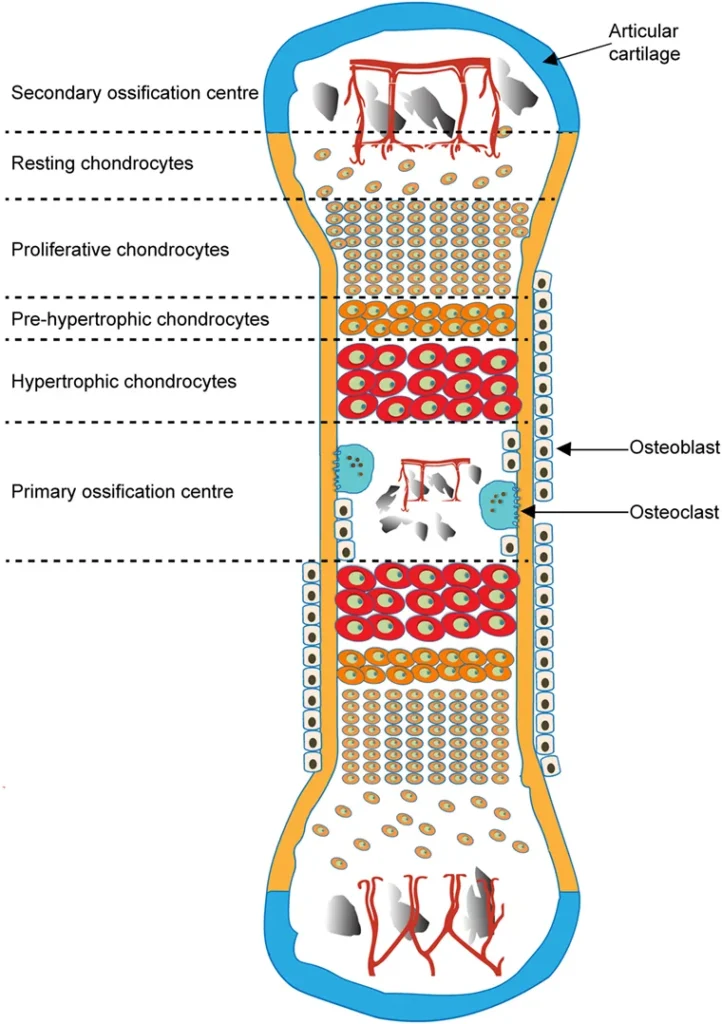
“Schematic of Endochondral Ossification and Formation of Primary and Secondary Ossification Centers” – Javaheri B., Caetano-Silva S.P., Kanakis I., Bou-Gharious G., Pitsillides A.A., via Wikimedia Commons.Licensed under CC BY 4.0.
Cartilage Ossification Replacement
Hypertrophic chondrocytes induce calcification of the surrounding cartilage matrix, which impairs nutrient diffusion and leads to chondrocyte apoptosis. The calcified cartilage serves as a temporary scaffold for subsequent bone deposition.
Invading blood vessels deliver osteoprogenitor cells, which differentiate into osteoblasts that deposit osteoid onto the remnants of calcified cartilage. This process establishes primary trabeculae, progressively replacing the cartilage framework with trabecular bone and marking the transition from cartilaginous to osseous tissue.
Development of Secondary Ossification Center
Following birth, secondary ossification centers develop within the epiphyses, initiating endochondral bone formation in these regions without formation of a medullary cavity. This process mirrors primary ossification but occurs independently at the bone ends.
A layer of hyaline cartilage persists between the epiphysis and metaphysis as the epiphyseal growth plate, which maintains longitudinal bone growth through coordinated chondrocyte proliferation, hypertrophy, and ossification.
These centers contribute to the formation of mature epiphyseal bone while preserving joint surfaces as articular cartilage, ensuring both growth potential and functional articulation.
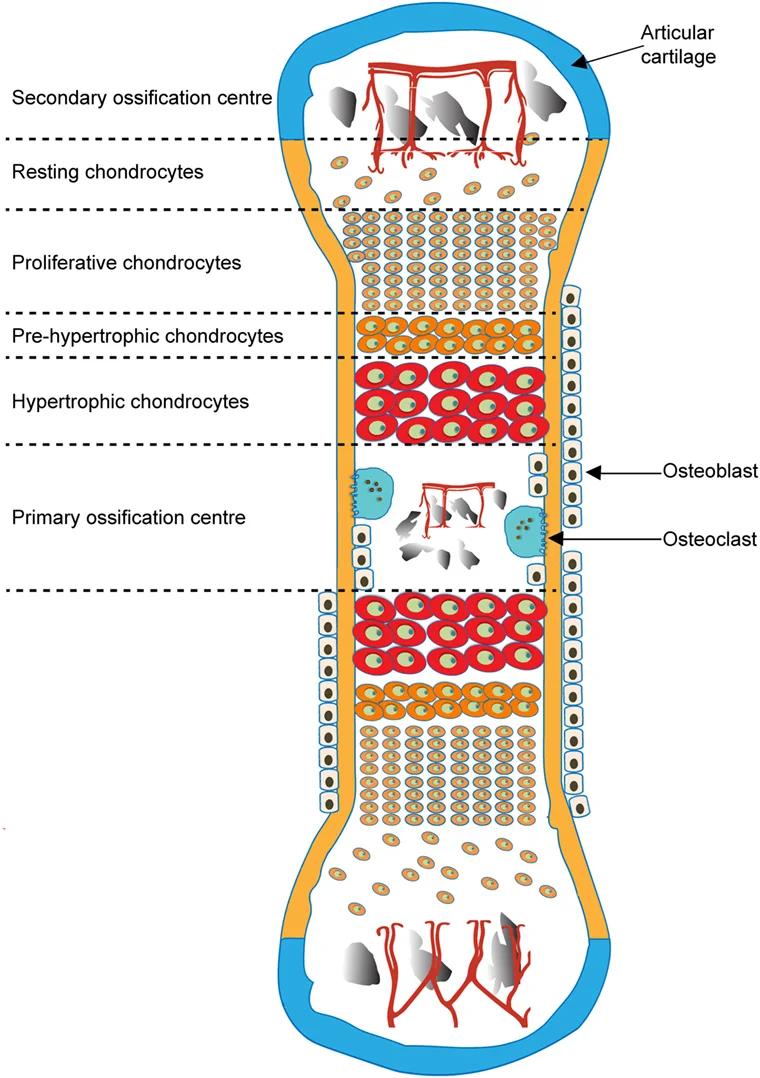
“Schematic of Endochondral Ossification and Formation of Primary and Secondary Ossification Centers” – Javaheri B., Caetano-Silva S.P., Kanakis I., Bou-Gharious G., Pitsillides A.A., via Wikimedia Commons.Licensed under CC BY 4.0.
Longitudinal Bone Growth
Long bones increase in length through activity of the epiphyseal growth plate, a specialized hyaline cartilage structure that enables controlled endochondral ossification via spatially organized zones of chondrocyte differentiation.
These zones include the resting (reserve) zone, containing quiescent progenitor cells; the proliferative zone, where chondrocytes undergo rapid mitosis and align in longitudinal columns; and the hypertrophic zone, characterized by cellular enlargement and matrix modification. This is followed by the zone of calcification, in which the cartilage matrix mineralizes, leading to chondrocyte apoptosis, and the zone of ossification, where invading blood vessels deliver osteoprogenitor cells that differentiate into osteoblasts and replace calcified cartilage with bone.
This coordinated progression results in continuous elongation of the diaphysis while maintaining the structural organization of the growth plate. Longitudinal growth persists until epiphyseal closure, when the cartilage plate is fully replaced by bone under the influence of hormonal regulation, particularly sex steroids.
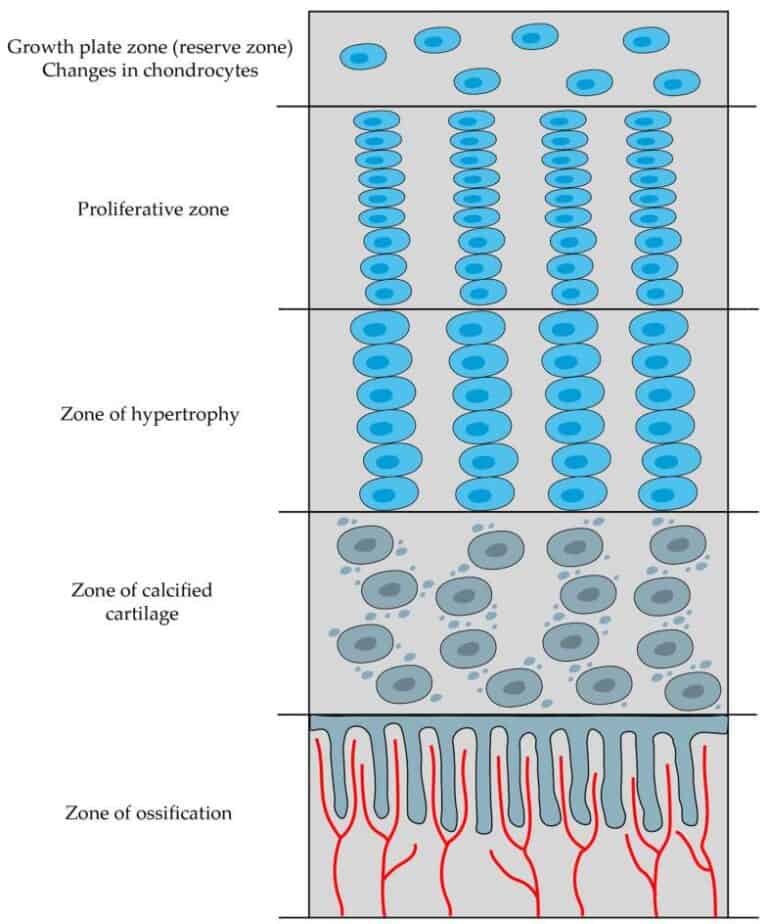
“Endochondral ossification and articular cartilage formation” by Zhangpeng Su et al., via NCBI/Wikimedia Commons. Licensed under CC BY SA 4.0
REGULATION OF OSSIFICATION
AI -generated illustration (MyoAantomy)
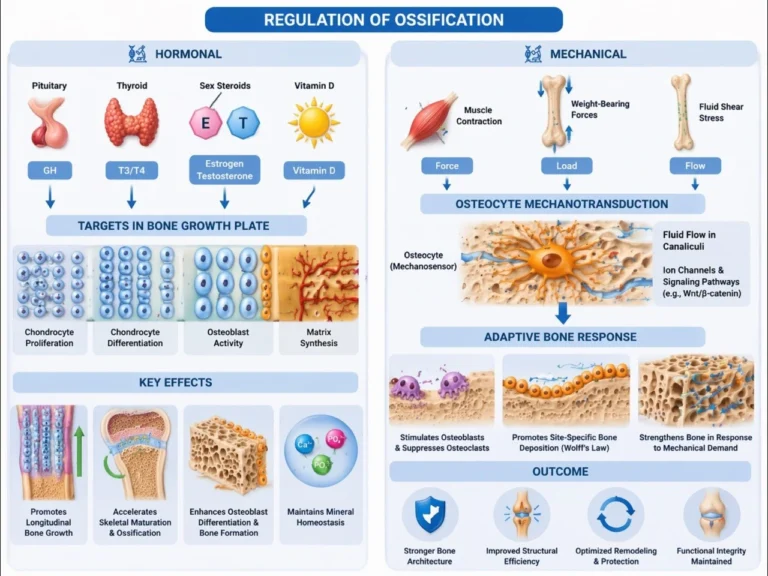
Hormonal
Skeletal development and bone remodeling are tightly regulated by endocrine signaling that coordinates chondrocyte activity, osteoblast function, and mineral homeostasis. Growth hormone (GH) stimulates longitudinal bone growth primarily via hepatic and local production of IGF-1, which promotes chondrocyte proliferation within the growth plate and enhances osteoblast differentiation and matrix synthesis.
Thyroid hormones (T3/T4) regulate the tempo of skeletal maturation by accelerating both chondrocyte differentiation and ossification processes. Sex steroids (estrogen and testosterone) exert dual effects: they initially enhance growth plate activity but subsequently induce epiphyseal closure through promotion of chondrocyte senescence and ossification.
Vitamin D maintains calcium and phosphate balance by increasing intestinal absorption and supporting mineral deposition in osteoid, ensuring proper matrix mineralization. Together, these hormonal systems integrate growth, maturation, and mineral homeostasis into a coordinated process of skeletal development
Exam Question
Explain how GH–IGF-1 axis, thyroid hormones, and sex steroids interact to regulate growth plate dynamics and determine the timing of epiphyseal closure.
Mechanical
Bone formation is dynamically regulated by mechanical loading through osteocyte-mediated mechanotransduction. Osteocytes function as primary mechanosensors, detecting strain induced by muscle contraction and weight-bearing forces, and translating it into biochemical signals that regulate bone remodeling.
Mechanical loading stimulates osteoblast activity and suppresses osteoclast-mediated resorption, promoting site-specific bone deposition along lines of stress (Wolff’s law). This adaptive remodeling optimizes bone architecture by reinforcing regions exposed to increased mechanical demand while preserving structural efficiency.
At the cellular level, mechanotransduction involves fluid flow within canaliculi, activation of ion channels, and signaling pathways (e.g., Wnt/β-catenin), which collectively regulate osteoblast differentiation and matrix production.
Exam Question
Describe the role of osteocytes in mechanotransduction and explain how mechanical loading influences the balance between bone formation and resorption to shape skeletal architecture.
FUNCTIONAL ROLE
Structural Framework
Ossification establishes the fundamental architectural and mechanical basis of the skeleton by transforming compliant connective tissues into rigid, mineralized bone.
This process creates the load-bearing framework required for posture, stability, and coordinated movement, while organizing cortical and trabecular bone into an optimized structure that balances strength, weight, and mechanical efficiency.
Force Transmission
Ossified bone functions as an effective lever system for skeletal muscles.
The rigidity achieved through mineralization enables efficient transmission of muscular forces via tendons across joints, allowing controlled movement. Simultaneously, the formation of reinforced entheses ensures safe transfer of tensile forces from muscles and ligaments to bone without structural failure.
Joint Formation
Ossification ensures the precise development of epiphyses and articular surfaces, which is essential for the formation of stable, congruent synovial joints.
Proper shaping of these surfaces allows efficient load distribution, minimizes friction during movement, and preserves long-term joint function and mechanical integrity.
Growth & Adaption
Through growth plate activity, ossification enables longitudinal bone growth and proportional development of the body.
Additionally, it provides the structural substrate for lifelong bone remodeling, allowing continuous adaptation to mechanical loading, maintenance of skeletal strength, and preservation of functional performance under changing physiological demands.
CLINICAL RLEVANCE
Achondroplasiaa
Achondroplasia is the most common form of disproportionate dwarfism and results from a mutation in the fibroblast growth factor receptor-3 (FGFR3) gene. This mutation inhibits chondrocyte proliferation within the epiphyseal growth plate, thereby impairing endochondral ossification.
As a result, longitudinal bone growth is reduced, producing shortened limbs while the axial skeleton remains relatively preserved.
Osteogesis Imperfectae
Osteogenesis imperfecta is a genetic disorder characterized by defective synthesis of type I collagen, the principal structural protein of bone matrix.
Because collagen provides the scaffold for mineral deposition during ossification, defective collagen formation leads to fragile bones with reduced mechanical strength. Clinically, this condition presents with recurrent fractures, skeletal deformities, and decreased bone density.
Rickets
Rickets is a disorder of defective bone mineralization, most commonly caused by vitamin D deficiency. Insufficient vitamin D impairs calcium and phosphate homeostasis, preventing proper mineral deposition within the osteoid matrix produced during ossification.
The resulting bones are soft and structurally weak, leading to skeletal deformities such as bowing of the long bones and widening of the growth plates.
Epiphyseal Injury
Trauma to the epiphyseal (growth) plates during childhood can disrupt the normal process of endochondral ossification responsible for longitudinal bone growth. Damage to the growth plate may lead to premature closure, asymmetric growth, or permanent limb length discrepancies, highlighting the critical role of growth plate integrity in skeletal development.
From a clinical perspective, understanding the mechanisms of ossification is essential in multiple medical disciplines, including orthopedics, pediatrics, endocrinology, and rehabilitation medicine. Accurate knowledge of these processes allows clinicians to diagnose developmental skeletal disorders, evaluate growth abnormalities, manage fractures involving growth plates, and develop therapeutic strategies aimed at restoring or preserving normal musculoskeletal function.
